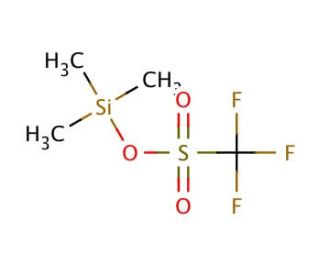
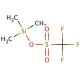
Trimethylsilyl trifluoromethanesulfonate (CAS 27607-77-8)
QUICK LINKS
Trimethylsilyl trifluoromethanesulfonate is a strong Lewis acid that functions as a highly effective catalyst in various chemical reactions. It acts as a source of the trifluoromethanesulfonate anion, which can be used to activate substrates for nucleophilic attack. This activation facilitates the formation of carbon-carbon and carbon-heteroatom bonds in organic synthesis. The mode of action involves the activation of electrophiles through coordination with the trifluoromethanesulfonate anion, leading to increased reactivity and selectivity in the desired chemical transformations. It serve as a promoter for the activation of silicon-containing compounds, enabling the generation of reactive intermediates for further applications. In this way, trimethylsilyl trifluoromethanesulfonate plays a role in facilitating the efficient and selective formation of complex organic molecules in research and development.
Trimethylsilyl trifluoromethanesulfonate (CAS 27607-77-8) References
- One-Pot Enol Silane Formation-Alkylation of Ketones with Propargyl Carboxylates Promoted by Trimethylsilyl Trifluoromethanesulfonate. | Downey, CW., et al. 2018. J Org Chem. 83: 12931-12938. PMID: 30248260
- Formal Enone α-Arylation via I(III)-Mediated Aryl Migration/Elimination. | Martins, BS., et al. 2021. Org Lett. 23: 2094-2098. PMID: 33635665
- Friedel-Crafts Addition of Indoles to Nitrones Promoted by Trimethylsilyl Trifluoromethanesulfonate. | Oracheff, ZZ., et al. 2021. J Org Chem. 86: 17328-17336. PMID: 34780184
- Contra-thermodynamic Olefin Isomerization by Chain-Walking Hydroboration and Dehydroboration. | Hanna, S., et al. 2022. Org Lett. 24: 1005-1010. PMID: 35080409
- Preparation of (Bis)Cationic Nitrogen-Ligated I(III) Reagents: Synthesis of [(pyridine)2IPh](OTf)2 and [(4-CF3-pyridine)2IPh](OTf)2. | Hoblos, B. and Wengryniuk, SE. 2021. Organic Synth. 98: 391-406. PMID: 35949486
- Synthesis of β,β-Disubstituted Styrenes via Trimethylsilyl Trifluoromethanesulfonate-Promoted Aldehyde-Aldehyde Aldol Coupling-Elimination. | Dixon, GJ., et al. 2022. J Org Chem. 87: 14846-14854. PMID: 36239694
- Enantioselective Total Syntheses of Preussomerins: Control of Spiroacetal Stereogenicity by Photochemical Reaction of a Naphthoquinone through 1,6-Hydrogen Atom Transfer. | Ando, Y., et al. 2023. Angew Chem Int Ed Engl. 62: e202213682. PMID: 36446739
- Adamantane-Substituted Purine Nucleosides: Synthesis, Host-Guest Complexes with β-Cyclodextrin and Biological Activity. | Rudolfová, J., et al. 2022. Int J Mol Sci. 23: PMID: 36499470
- Design and Synthesis of Neutralizable Fondaparinux. | Zhang, L., et al. 2022. JACS Au. 2: 2791-2799. PMID: 36590263
- Silylium-Catalyzed Regio- and Stereoselective Carbosilylation of Ynamides with Allylic Trimethylsilanes. | Yepes, P., et al. 2023. Org Lett. 25: 1020-1024. PMID: 36749888
- Design and Synthesis of New Modified Flexible Purine Bases as Potential Inhibitors of Human PNP. | Khandazhinskaya, A., et al. 2023. Molecules. 28: PMID: 36770593
- Continuous-Flow Synthesis of Δ9-Tetrahydrocannabinol and Δ8-Tetrahydrocannabinol from Cannabidiol. | Bassetti, B., et al. 2023. J Org Chem. 88: 6227-6231. PMID: 37014222
- A convergent fragment coupling strategy to access quaternary stereogenic centers. | Kerkovius, JK., et al. 2023. Chem Sci. 14: 4397-4400. PMID: 37123185
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Trimethylsilyl trifluoromethanesulfonate, 25 g | sc-255700 | 25 g | $47.00 | |||
Trimethylsilyl trifluoromethanesulfonate, 50 g | sc-255700A | 50 g | $164.00 |
