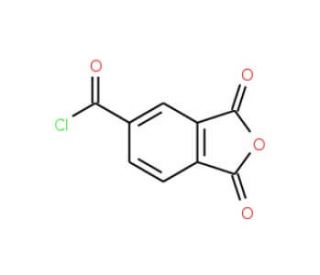
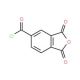
Trimellitic anhydride chloride (CAS 1204-28-0)
QUICK LINKS
Trimellitic anhydride chloride, a member of the cyclic anhydrides family, is a chloride derivative of trimellitic acid—a benzenetricarboxylic acid. This compound presents as a colorless to pale yellow crystalline solid, dissolving well in organic solvents such as acetone, benzene, and chloroform. It plays a significant role as a monomer in crafting polyamide resins and polyurethane elastomers. When combined with other monomers like methyl acrylate and ethyl acrylate, it aids in the formation of various polymers. An important characteristic to note is its reaction with water, which produces hydrogen chloride gas—a factor that can lead to metal corrosion. Moreover, upon exposure to water vapor, it forms hydrochloric acid and water.
Trimellitic anhydride chloride (CAS 1204-28-0) References
- Synthesis and antimicrobial activity of new adamantane derivatives I. | Orzeszko, A., et al. 2000. Acta Biochim Pol. 47: 87-94. PMID: 10961681
- Synthesis and antimicrobial activity of new adamantane derivatives II. | Orzeszko, A., et al. 2000. Farmaco. 55: 619-23. PMID: 11152243
- Synthesis of new silane coupling agents with a trimellitic anhydride group and application as primers for ceramics and alloys. | Kurata, S. and Umemoto, K. 2007. Dent Mater J. 26: 800-4. PMID: 18203484
- Inhibition of carbonic anhydrase isoforms I, II, IV, VII and XII with carboxylates and sulfonamides incorporating phthalimide/phthalic anhydride scaffolds. | El-Azab, AS., et al. 2016. Bioorg Med Chem. 24: 20-5. PMID: 26678172
- Facile synthesis of AIE-active amphiphilic polymers: Self-assembly and biological imaging applications. | Long, Z., et al. 2016. Mater Sci Eng C Mater Biol Appl. 66: 215-220. PMID: 27207057
- Nature and kinetics of redox imbalance triggered by respiratory and skin chemical sensitizers on the human monocytic cell line THP-1. | Ferreira, I., et al. 2018. Redox Biol. 16: 75-86. PMID: 29477863
- Synthesis and characterization of novel trimellitic anhydride isothiocyanate-cross linked chitosan hydrogels modified with multi-walled carbon nanotubes for enhancement of antimicrobial activity. | Mohamed, NA., et al. 2019. Int J Biol Macromol. 132: 416-428. PMID: 30930265
- Blocking of CD4 cell receptors for the human immunodeficiency virus type 1 (HIV-1) by chemically modified bovine milk proteins: potential for AIDS prophylaxis. | Neurath, AR., et al. 1995. J Mol Recognit. 8: 304-16. PMID: 8619951
- Versatile linker chemistry for synthesis of 3'-modified DNA. | Lyttle, MH., et al. 1997. Bioconjug Chem. 8: 193-8. PMID: 9095360
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Trimellitic anhydride chloride, 25 g | sc-280154 | 25 g | $64.00 |
