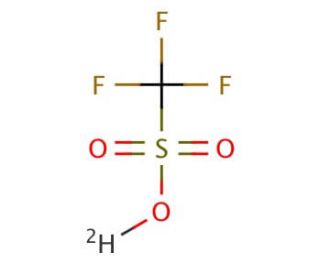
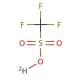
Trifluoromethanesulfonic acid-d (CAS 66178-41-4)
QUICK LINKS
Trifluoromethanesulfonic acid-d is a fluorinated sulfonate for proteomics research. Trifluoromethanesulfonic acid-d is a strong acid known for its reactivity with nucleophiles such as water, alcohols, and amines. It is used in organic synthesis, particularly in desulfurization reactions, enabling the conversion of thiophene to benzothiophene. To obtain Trifluoromethanesulfonic acid-d, a synthetic chemical, the process involves the reaction between sulfur trioxide and chlorotrifluoroethanesulfonyl fluoride. The protonation of Trifluoromethanesulfonic acid-d molecule generates a sulfide anion, which then reacts with a nucleophile, such as a chloride ion, resulting in the generation of a chloride anion and the regeneration of Trifluoromethanesulfonic acid-d molecule. This cyclic process allows Trifluoromethanesulfonic acid-d to react again with another nucleophile.
Trifluoromethanesulfonic acid-d (CAS 66178-41-4) References
- Sequential protonation and methylation of a hydride-osmium complex containing a cyclopentadienyl ligand with a pendant amine group. | Esteruelas, MA., et al. 2005. Inorg Chem. 44: 4094-103. PMID: 15907139
- Hydrogen/deuterium exchange of cross-linkable α-amino acid derivatives in deuterated triflic acid. | Wang, L., et al. 2014. Biosci Biotechnol Biochem. 78: 1129-34. PMID: 25229846
- Proton self‐exchange of para‐substituted N, N‐dimethylanilines and their conjugate acids in aprotic solvents | Goez, M., & Heun, R. 1998. Berichte der Bunsengesellschaft für physikalische Chemie. 102(3): 560-566.
- Metal-stabilized methylene arenium and σ-arenium compounds: Synthesis, structure, reactivity, charge distribution, and interconversion | Vigalok, A., Rybtchinski, B., Shimon, L. J., Ben-David, Y., & Milstein, D. 1999. Organometallics. 18(5): 895-905.
- Spontaneous oxidation of organic donors to their cation radicals using Brønsted acids. Identification of the elusive oxidant | Rajendra Rathore, Chengjian Zhu, Sergey V. Lindemana and Jay K. Kochi. 2000. Journal of the Chemical Society, Perkin Transactions 2. (9): 1837-1840.
- Observation of quantum paraelectricity in an intermolecular ionic hydrogen-bonded crystal of a squaric acid derivative | Takasu, I., Izuoka, A., Sugawara, T., & Mochida, T. 2004. The Journal of Physical Chemistry B. 108(18): 5527-5531.
- Novel diapocarotenoid dications with VIS/NIR absorption | Kildahl-Andersen, G., Anthonsen, T., & Liaaen-Jensen, S. 2006. Tetrahedron letters. 47(27): 4693-4696.
- Formation of 1, 4-diphosphinobenzenes via tele-substitution on fluorobenzenechromium complexes | Yamamoto, Y., Danjo, H., Yamaguchi, K., & Imamoto, T. 2008. Journal of Organometallic Chemistry. 693(23): 3546-3552.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Trifluoromethanesulfonic acid-d, 1 g | sc-258297 | 1 g | $43.00 | |||
Trifluoromethanesulfonic acid-d, 5 g | sc-258297A | 5 g | $141.00 | |||
Trifluoromethanesulfonic acid-d, 10 g | sc-258297B | 10 g | $235.00 |
