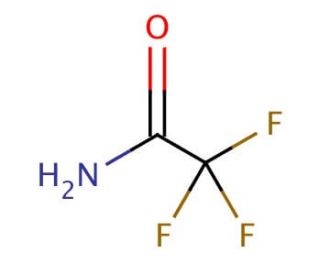
Trifluoroacetamide (CAS 354-38-1)
QUICK LINKS
Trifluoroacetamide is a compound that functions as a derivatizing agent in analytical chemistry. It is used to derivatize carboxylic acids and amines for analysis by gas chromatography and mass spectrometry. It may react with the functional groups of the target compounds, forming stable derivatives that can be easily analyzed. The mode of action involves the substitution of the hydrogen atoms in the carboxylic acids and amines with trifluoroacetamide groups, resulting in the formation of derivatives with improved volatility and stability. This allows for enhanced detection and quantification of the target compounds in complex mixtures. Trifluoroacetamide′s role in this process is to facilitate the analysis of carboxylic acids and amines, providing access to important insights about the composition of samples in various experimental applications.
Trifluoroacetamide (CAS 354-38-1) References
- Electrochemistry in deep eutectic solvents. | Nkuku, CA. and LeSuer, RJ. 2007. J Phys Chem B. 111: 13271-7. PMID: 17973421
- Catalytic asymmetric C-N bond formation: phosphine-catalyzed intra- and intermolecular γ-addition of nitrogen nucleophiles to allenoates and alkynoates. | Lundgren, RJ., et al. 2013. Angew Chem Int Ed Engl. 52: 2525-8. PMID: 23339132
- Determination of membrane potential and cell volume by 19F NMR using trifluoroacetate and trifluoroacetamide probes. | London, RE. and Gabel, SA. 1989. Biochemistry. 28: 2378-82. PMID: 2730869
- Oxazoline-Promoted Rh-Catalyzed C-H Amidation of Benzene Derivatives with Sulfonamides and Trifluoroacetamide. A Comparative Study. | Maiden, TM., et al. 2016. J Org Chem. 81: 10641-10650. PMID: 27807971
- Rhodium-catalyzed regioselective C8-H amination of quinoline N-oxides with trifluoroacetamide at room temperature. | You, C., et al. 2018. Org Biomol Chem. 16: 4728-4733. PMID: 29901064
- Synthesis of Fused Bicyclic [1,2,3]-Triazoles from γ-Amino Diazoketones. | Santiago, JV. and Burtoloso, ACB. 2019. ACS Omega. 4: 159-168. PMID: 31459321
- Close Amide NH···F Hydrogen Bonding Interactions in 1,8-Disubstituted Naphthalenes. | Kazim, M., et al. 2020. J Org Chem. 85: 6195-6200. PMID: 32227992
- Deep Eutectic Solvent Based on Lithium Bis[(trifluoromethyl)sulfonyl] Imide (LiTFSI) and 2,2,2-Trifluoroacetamide (TFA) as a Promising Electrolyte for a High Voltage Lithium-Ion Battery with a LiMn2O4 Cathode. | Dinh, TTA., et al. 2020. ACS Omega. 5: 23843-23853. PMID: 32984704
- Photocatalytic defluoroalkylation and hydrodefluorination of trifluoromethyls using o-phosphinophenolate. | Liu, C., et al. 2022. Nat Commun. 13: 354. PMID: 35039496
- Electrophotocatalytic oxygenation of multiple adjacent C-H bonds. | Shen, T., et al. 2023. Nature. 614: 275-280. PMID: 36473497
- Measurement of the diffusion of 2,2,2-trifluoroacetamide within thermoresponsive hydrogels using NMR imaging. | Dinarvand, R., et al. 1995. Pharm Res. 12: 1376-9. PMID: 8570538
- Copper-catalyzed synthesis of primary arylamines from aryl halides and 2,2,2-trifluoroacetamide | Tao, C. Z., Li, J., Fu, Y., Liu, L., & Guo, Q. X. 2008. Tetrahedron Letters. 49(1): 70-75.
- Elimination of All Free Glycerol and Reduction of Total Glycerol from Palm Oil-Based Biodiesel Using Non-Glycerol Based Deep Eutectic Solvents | Shahbaz, K., Mjalli, F. S., Hashim, M. A., & AlNashef, I. M. 2013. Separation Science and Technology. 48(8): 1184-1193.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Trifluoroacetamide, 25 g | sc-251333 | 25 g | $37.00 | |||
Trifluoroacetamide, 250 g | sc-251333A | 250 g | $50.00 | |||
Trifluoroacetamide, 1 kg | sc-251333B | 1 kg | $170.00 | |||
Trifluoroacetamide, 2.5 kg | sc-251333C | 2.5 kg | $380.00 | |||
Trifluoroacetamide, 5 kg | sc-251333D | 5 kg | $720.00 | |||
Trifluoroacetamide, 10 kg | sc-251333E | 10 kg | $1400.00 |
