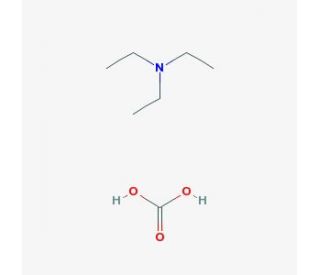
Molecular structure of Triethylammonium bicarbonate buffer, CAS Number: 15715-58-9
Triethylammonium bicarbonate buffer (CAS 15715-58-9)
Alternate Names:
Buffer solution 1 M pH 8.4-8.6 (volatile); Triethylammonium hydrogen carbonate buffer
Application:
Triethylammonium bicarbonate buffer is a buffer for ion-exchange chromatography
CAS Number:
15715-58-9
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Triethylammonium bicarbonate buffer is a buffer used in ion-exchange chromatography and electrophoresis. Triethylammonium bicarbonate buffer is widely used in scientific research for a variety of purposes. It is used as a buffer in aqueous solutions to maintain a constant pH, and it is also used as a stabilizer in enzyme reactions.
Triethylammonium bicarbonate buffer (CAS 15715-58-9) References
- Stabilization of gas-phase noncovalent macromolecular complexes in electrospray mass spectrometry using aqueous triethylammonium bicarbonate buffer. | Lemaire, D., et al. 2001. Anal Chem. 73: 1699-706. PMID: 11338582
- Isolation and characterization of 2-thiocytidine from a serine transfer ribonucleic acid of Escherichia coli. | Yamada, Y., et al. 1970. FEBS Lett. 7: 207-210. PMID: 11947472
- ISOLATION AND QUANTITATIVE DETERMINATION OF PSEUDOURIDINE IN URINE BY ANION-EXCHANGE CHROMATOGRAPHY. | RINDERKNECHT, H. and MA, V. 1964. J Chromatogr. 16: 407-9. PMID: 14245477
- Photochemistry and thermal decarboxylation of alpha-phosphoryloxy-p-nitrophenylacetates. | Corrie, JE., et al. 2009. Photochem Photobiol. 85: 1089-96. PMID: 19496988
- Dimensional probes of the enzyme binding sites of adenine nucleotides. Biological effects of widening the adenine ring by 2.4 A. | Leonard, NJ., et al. 1978. Biochemistry. 17: 3677-85. PMID: 212100
- An integrated workflow for extraction and solubilization of intermediate filaments from colorectal biopsies for proteomic analysis. | Majumdar, D., et al. 2012. Electrophoresis. 33: 1967-74. PMID: 22806461
- Phosphoenolpyruvate synthetase and pyruvate, orthophosphate dikinase: stereochemical consequences at both the beta-phospho and gamma-phospho groups of ATP. | Cook, AG. and Knowles, JR. 1985. Biochemistry. 24: 51-8. PMID: 2986676
- Evaluation and minimization of nonspecific tryptic cleavages in proteomic sample preparation. | Lin, Z., et al. 2020. Rapid Commun Mass Spectrom. 34: e8733. PMID: 32031715
- Separation of nucleosides and nucleotides by reversed-phase high-performance liquid chromatography with volatile buffers allowing sample recovery. | Ip, CY., et al. 1985. Anal Biochem. 147: 180-5. PMID: 4025816
- Dentin phosphoprotein: an extracellular calcium-binding protein. | Lee, SL., et al. 1977. Biochemistry. 16: 2971-9. PMID: 406911
- Nucleosides and nucleotides. VI. Preparation of diribonucleoside monophosphates containing 4-thiouridine. | Miura, K., et al. 1973. J Biochem. 73: 1279-84. PMID: 4724303
- The stereochemical course of nucleotidyl transfer catalyzed by ATP sulfurylase. | Bicknell, R., et al. 1982. J Biol Chem. 257: 8922-7. PMID: 6284750
- A stereochemical and positional isotope-exchange study of the mechanism of activation of methionine by methionyl-tRNA synthetase from Escherichia coli. | Lowe, G., et al. 1983. Eur J Biochem. 130: 341-5. PMID: 6337846
- An investigation of the mechanism of activation of tryptophan by tryptophanyl-tRNA synthetase from beef pancreas. | Lowe, G. and Tansley, G. 1984. Eur J Biochem. 138: 597-602. PMID: 6692836
- Natural and synthetic non-peptide antigens recognized by human gamma delta T cells. | Tanaka, Y., et al. 1995. Nature. 375: 155-8. PMID: 7753173
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Triethylammonium bicarbonate buffer, 100 ml | sc-253749 | 100 ml | $163.00 | |||
Triethylammonium bicarbonate buffer, 500 ml | sc-253749A | 500 ml | $490.00 | |||
Triethylammonium bicarbonate buffer, 1 L | sc-253749B | 1 L | $918.00 |
