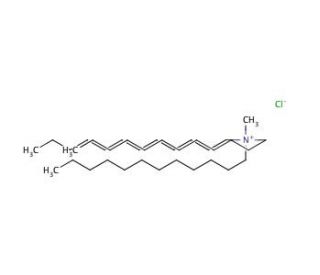
Molecular structure of Tridodecylmethylammonium chloride, CAS Number: 7173-54-8
Tridodecylmethylammonium chloride (CAS 7173-54-8)
Alternate Names:
Methyltridodecylammonium chloride
CAS Number:
7173-54-8
Purity:
≥95%
Molecular Weight:
572.47
Molecular Formula:
C37H78ClN
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Tridodecylmethylammonium chloride (TDMAC) is a quaternary ammonium salt extensively utilized in diverse scientific and industrial fields. It serves as a cationic surfactant produced by the reaction between tridodecylmethylamine and hydrochloric acid. Tridodecylmethylammonium chloride functions by reducing the surface tension of water, acting as a surfactant. It also acts as an emulsifier, facilitating the creation of stable emulsions. Due to its cationic properties, it can effectively bind with anionic substances like proteins, forming stable complexes. Moreover, Tridodecylmethylammonium chloride exhibits antifungal properties by disrupting the cell membrane of fungi.
Tridodecylmethylammonium chloride (CAS 7173-54-8) References
- Mercuracarborand 'anti-crown ether'-based chloride-sensitive liquid/polymeric membrane electrodes. | Badr, IH., et al. 1999. Anal Chem. 71: 1371-7. PMID: 10204038
- Rotating electrode potentiometry: lowering the detection limits of nonequilibrium polyion-sensitive membrane electrodes. | Ye, Q. and Meyerhoff, ME. 2001. Anal Chem. 73: 332-6. PMID: 11199986
- The influence of CO(2) on ISFETs with polymer membranes and characterization of a carbonate ion sensor. | Abramova, N., et al. 2010. Talanta. 81: 1750-4. PMID: 20441968
- Platinum porphyrins as ionophores in polymeric membrane electrodes. | Lvova, L., et al. 2011. Analyst. 136: 4966-76. PMID: 21971176
- Direct optical carbon dioxide sensing based on a polymeric film doped with a selective molecular tweezer-type ionophore. | Xie, X., et al. 2012. Anal Chem. 84: 3163-9. PMID: 22369632
- Osmium-191/iridium-191m generator based on silica gel impregnated with tridodecylmethylammonium chloride. | Issachar, D., et al. 1989. J Nucl Med. 30: 538-41. PMID: 2544695
- Ionophore-based potentiometric PVC membrane sensors for determination of phenobarbitone in pharmaceutical formulations. | Alrabiah, H., et al. 2016. Acta Pharm. 66: 503-514. PMID: 27749249
- Characterization and Quantification of Polyquaterniums via Single-Use Polymer Membrane-Based Polyion-Sensitive Electrodes. | Ferguson, SA. and Meyerhoff, ME. 2017. ACS Sens. 2: 268-273. PMID: 28723131
- Reduction of bacterial adherence to catheter surface with heparin. | Ruggieri, MR., et al. 1987. J Urol. 138: 423-6. PMID: 3298698
- A colorimetric paper-based optode sensor for highly sensitive and selective determination of thiocyanate in urine sample using cobalt porphyrin derivative. | Wirojsaengthong, S., et al. 2021. Talanta. 231: 122371. PMID: 33965036
- Antibiotic bonding to polytetrafluoroethylene with tridodecylmethylammonium chloride. | Harvey, RA., et al. 1982. Surgery. 92: 504-12. PMID: 7112401
- A novel electrochemical heparin sensor. | Yang, VC., et al. 1993. ASAIO J. 39: M195-201. PMID: 8268527
- Electrochemical sensor for heparin: further characterization and bioanalytical applications. | Ma, SC., et al. 1993. Anal Chem. 65: 2078-84. PMID: 8372971
- A disposable, coated wire heparin sensor. | Yun, JH., et al. 1994. ASAIO J. 40: M401-5. PMID: 8555546
- Clinical application of disposable heparin sensors. Blood heparin measurements during open heart surgery. | Yun, JH., et al. 1995. ASAIO J. 41: M661-4. PMID: 8573887
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tridodecylmethylammonium chloride, 1 g | sc-255693 | 1 g | $173.00 |
