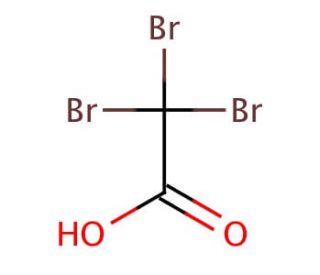
Tribromoacetic acid (CAS 75-96-7)
QUICK LINKS
Tribromoacetic acid is frequently employed in organic chemistry research, particularly in the study of halogenated compounds and their reactivity. This acid is used as a reagent in the bromination of various organic substrates, where its high electrophilic character enables selective transformations. In synthetic chemistry, Tribromoacetic acid is explored for its utility in the synthesis of brominated organic molecules, which are important for further chemical modifications or as building blocks in more complex chemical structures. Tribromoacetic acid is utilized in environmental chemistry to investigate the fate and transformation of brominated organic compounds in natural waters and soils. Researchers also study the mechanisms by which Tribromoacetic acid interacts with other chemical species, which is for understanding its behavior in both controlled and natural environments.
Tribromoacetic acid (CAS 75-96-7) References
- New Perspectives for Cancer Hazard Evaluation by the Report on Carcinogens: A Case Study Using Read-Across Methods in the Evaluation of Haloacetic Acids Found as Water Disinfection By-Products. | Atwood, ST., et al. 2019. Environ Health Perspect. 127: 125003. PMID: 31854200
- Simultaneous Determination of Chlorinated and Brominated Acetic Acids in Various Environmental Water Matrixes by High-Performance Liquid Chromatography-Inductively Coupled Plasma Tandem Mass Spectrometry without Sample Preparation. | Lajin, B. and Goessler, W. 2020. Anal Chem. 92: 9156-9163. PMID: 32545952
- Insights into unexpected photoisomerization from photooxidation of tribromoacetic acid in aqueous environment using ultrafast spectroscopy. | Yeung, CS., et al. 2021. J Hazard Mater. 418: 126214. PMID: 34102359
- Ecological impacts of ballast water loading and discharge: insight into the toxicity and accumulation of disinfection by-products. | Kurniawan, SB., et al. 2022. Heliyon. 8: e09107. PMID: 35309395
- [Determination of Haloacetic Acids, Disinfection Byproducts, in Tap Water with Reversed-Phase Ultra-Performance Liquid Chromatography-High Resolution Mass Spectrometry]. | Zhao, X., et al. 2022. Sichuan Da Xue Xue Bao Yi Xue Ban. 53: 504-510. PMID: 35642162
- Dissipative Formation of Covalent Basket Cages. | Liyana Gunawardana, VW., et al. 2022. Angew Chem Int Ed Engl. 61: e202207418. PMID: 35723284
- Disinfection byproducts in chlorinated or brominated swimming pools and spas: Role of brominated DBPs and association with mutagenicity. | Liberatore, HK., et al. 2022. J Environ Sci (China). 117: 253-263. PMID: 35725077
- Insights to estimate exposure to regulated and non-regulated disinfection by-products in drinking water. | Redondo-Hasselerharm, PE., et al. 2022. J Expo Sci Environ Epidemiol. 1-11. PMID: 35768489
- Cancer Hazard Evaluations for Contemporary Needs: Highlights From New National Toxicology Program Evaluations and Methodological Advancements. | Lunn, RM., et al. 2022. J Natl Cancer Inst. 114: 1441-1448. PMID: 36029241
- Toxicity evaluation of chlorinated natural water using Photobacterium phosphoreum: Implications for ballast water management. | Yang, M., et al. 2023. J Environ Manage. 335: 117471. PMID: 36863148
- Dissipative Systems Driven by the Decarboxylation of Activated Carboxylic Acids. | Del Giudice, D. and Di Stefano, S. 2023. Acc Chem Res. 56: 889-899. PMID: 36916734
- Genotoxicity and endocrine disruption potential of haloacetic acids in human placental and lung cells. | Pérez-Albaladejo, E., et al. 2023. Sci Total Environ. 879: 162981. PMID: 36963690
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tribromoacetic acid, 1 g | sc-258282 | 1 g | $48.00 |
