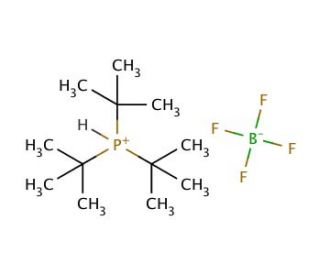
Tri-tert-butylphosphonium tetrafluoroborate (CAS 131274-22-1)
QUICK LINKS
Tri-tert-butylphosphonium tetrafluoroborate is extensively studied in the field of ionic liquids and phase transfer catalysis. This compound is particularly valued for its ability to facilitate the transfer of anions across different phases, which is for reactions that require the efficient movement of ions from aqueous to organic layers. Research on Tri-tert-butylphosphonium tetrafluoroborate includes its application in organic synthesis, where it acts as a catalyst to enhance the rate and selectivity of chemical reactions. The stability and solubility of this compound in various solvents are also key topics of investigation, influencing its effectiveness and applicability in different chemical environments. Furthermore, studies focus on its role in green chemistry, where it is used to develop more environmentally friendly synthetic pathways by reducing solvent use and enhancing reaction efficiencies.
Tri-tert-butylphosphonium tetrafluoroborate (CAS 131274-22-1) References
- Rhodium-catalyzed tandem cyclization: formation of 1H-indenes and 1-alkylideneindans from arylboronate esters in aqueous media. | Lautens, M. and Marquardt, T. 2004. J Org Chem. 69: 4607-14. PMID: 15230582
- Palladium catalysts for highly selective Sonogashira reactions of aryl and heteroaryl bromides. | Torborg, C., et al. 2008. ChemSusChem. 1: 91-6. PMID: 18605670
- Palladium-catalyzed cross-coupling of cyclopropylmagnesium bromide with aryl bromides mediated by zinc halide additives. | Shu, C., et al. 2010. J Org Chem. 75: 6677-80. PMID: 20815373
- Mechanistic analysis of azine N-oxide direct arylation: evidence for a critical role of acetate in the Pd(OAc)2 precatalyst. | Sun, HY., et al. 2010. J Org Chem. 75: 8180-9. PMID: 21053903
- Synthesis of Intricate Fused N-Heterocycles via Ring-Rearrangement Metathesis. | Kotha, S. and Gunta, R. 2017. J Org Chem. 82: 8527-8535. PMID: 28721727
- 5-Bromo-norborn-2-en-7-one derivatives as a carbon monoxide source for palladium catalyzed carbonylation reactions. | Payne, CM., et al. 2019. RSC Adv. 9: 30736-30740. PMID: 35529407
- Palladium-catalyzed solid-state borylation of aryl halides using mechanochemistry. | Kubota, K., et al. 2022. Beilstein J Org Chem. 18: 855-862. PMID: 35957749
- Microwave assisted, palladium catalyzed aminocarbonylations of heteroaromatic bromides using solid Mo(CO)6 as the carbon monoxide source | Michael A. Letavic, Kiev S. Ly. 2007. Tetrahedron Letters. 48: 2339-2343.
- Straightforward access to aryl-substituted tetrathiafulvalenes by palladium-catalysed direct C–H arylation and their photophysical and electrochemical properties | Yukihiro Mitamura ab, Hideki Yorimitsu *a, Koichiro Oshima *bc and Atsuhiro Osuka a. 2011. Chem. Sci. 2: 2017-2021.
- Synthesis of new n-type isoindigo copolymers† | François Grenier,a Philippe Berrouard,ab Jean-Rémi Pouliot,a Hsin-Rong Tseng,b Alan J. Heegerb and Mario Leclerc*a. 2013. Polym. Chem. 4: 1836-1841.
- 11C-Carbonylation through in Situ Generated 11C-Benzoyl Chlorides with Tetrabutylammonium Chloride as Chloride Source | Kenneth Dahl, Patrik Nordeman. 2017. European Journal of Organic Chemistry. 2017: 2648-2651.
- Designing highly luminescent aryloxy-benzothiadiazole derivatives with aggregation-induced enhanced emission | Alessandra Pazini a, Luis Maqueira a, Fabiano da Silveira Santos b, Arthur Rodrigues Jardim Barreto c, Rafael dos Santos Carvalho c, Felipe Miranda Valente a, Davi Back d, Ricardo Queiroz Aucélio a, Marco Cremona c, Fabiano Severo Rodembusch b, Jones Limberger a. 2020. Dyes and Pigments. 178.
- Synthesis of push–pull triarylamine dyes containing 5,6-difluoro-2,1,3-benzothiadiazole units by direct arylation and their evaluation as active material for organic photovoltaics | Tatiana Ghanem a, Tony Vincendeau a, Pablo Simón Marqués a, Amir Hossein Habibi a, Sana Abidi ab, Ali Yassin a, Sylvie Dabos-Seignon a, Jean Roncali c, Philippe Blanchard a and Clément Cabanetos *ad. 2021. Mater. Adv. 2: 7456-7462.
- A new V-shaped 2H-imidazole-based spirocyclic fluorophore: Aggregation-induced emission, twisted intramolecular charge transfer, and high responsiveness to trace water and acid | Feiyue Du a 1, Dongqin Li b 1, Sheng Ge a, Shumei Xie a, Mi Tang a, Ziqiang Xu a, Erjing Wang a, Shimin Wang a, Ben Zhong Tang c. 2021. Dyes and Pigments. 194.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tri-tert-butylphosphonium tetrafluoroborate, 1 g | sc-253733 | 1 g | $85.00 |
