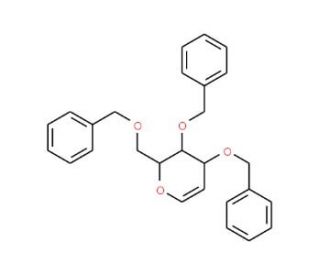
Tri-O-benzyl-D-glucal (CAS 55628-54-1)
QUICK LINKS
Tri-O-benzyl-D-glucal is a protected form of glucal, a derivative of glucose, where three hydroxyl groups are benzylated, enhancing its stability and reactivity in organic synthesis. This compound is widely used in carbohydrate chemistry, particularly in the synthesis of glycosides and oligosaccharides. Its benzyl groups serve as protecting groups that can be selectively removed under mild conditions, allowing for controlled glycosylation reactions. In research, Tri-O-benzyl-D-glucal is often utilized as a glycosyl donor in the synthesis of complex carbohydrates. It undergoes glycosylation reactions facilitated by Lewis acids or other catalysts, forming glycosidic bonds essential for constructing carbohydrate chains. This makes it invaluable in studying the formation and function of glycosidic linkages in biological systems. Additionally, Tri-O-benzyl-D-glucal is used to investigate the mechanisms of glycosyltransferase enzymes, which are crucial for the biosynthesis of glycoproteins and glycolipids. Researchers also employ this compound in the synthesis of carbohydrate-based ligands and probes, aiding in the exploration of carbohydrate-protein interactions and cell surface recognition processes. Overall, Tri-O-benzyl-D-glucal is a critical tool for advancing our understanding of carbohydrate synthesis, enzyme mechanisms, and the role of glycoconjugates in biological systems.
Tri-O-benzyl-D-glucal (CAS 55628-54-1) References
- Substituent dependence of the diastereofacial selectivity in iodination and bromination of glycals and related cyclic enol ethers. | Boschi, A., et al. 2000. J Org Chem. 65: 8470-7. PMID: 11112566
- Acetyl Perchlorate Mediated Rearrangement of Tri-O-benzyl-D-glucal. Evidence for a 1,6-Hydride Shift. | Byerley, AL., et al. 1998. J Org Chem. 63: 193-194. PMID: 11674063
- One-pot synthesis of C-glycosylic compounds (C-glycosides) from D-glucal, p-tolylsulfenyl chloride and aromatic/heteroaromatic compounds in the presence of Lewis acids. | Koikov, LN., et al. 2002. Carbohydr Res. 337: 1275-83. PMID: 12151207
- General synthesis and biological evaluation of alpha-1-C-substituted derivatives of fagomine (2-deoxynojirimycin-alpha-C-glycosides). | Goujon, JY., et al. 2005. Bioorg Med Chem. 13: 2313-24. PMID: 15727880
- Design and synthesis of new amino-modified iminocyclitols: selective inhibitors of alpha-galactosidase. | Ganesan, M., et al. 2010. Org Biomol Chem. 8: 1527-30. PMID: 20237662
- Synthesis of C-furanosides from a D-glucal-derived cyclopropane through a ring-expansion/ring-contraction sequence. | Hewitt, RJ. and Harvey, JE. 2011. Chem Commun (Camb). 47: 421-3. PMID: 20852781
- A Chiron Approach to Diversity-Oriented Synthesis of Aminocyclitols, (-)-Conduramine F-4 and Polyhydroxyaminoazepanes from a Common Precursor. | Harit, VK. and Ramesh, NG. 2016. J Org Chem. 81: 11574-11586. PMID: 27806198
- Ring closing metathesis (RCM) approach to the synthesis of conduramine B-2, ent-conduramine F-2, aminocyclopentitol and trihydroxyazepane. | Harit, VK. and Ramesh, NG. 2019. Org Biomol Chem. 17: 5951-5961. PMID: 31166343
- An expeditious synthesis of novel DNA nucleobase mimics of (+)-anisomycin. | Mishra, UK., et al. 2022. Carbohydr Res. 520: 108645. PMID: 35964481
- Rational design and synthesis of novel triazole- and tetrazole-fused iminosugars as potential inhibitors of amyotrophic lateral sclerosis (ALS) linked SOD1 aggregation. | Santhanam, V., et al. 2023. Int J Biol Macromol. 253: 126900. PMID: 37714236
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tri-O-benzyl-D-glucal, 5 g | sc-258280 | 5 g | $163.00 |
