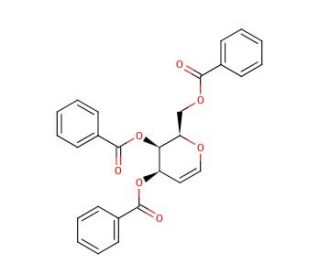
Tri-O-benzoyl-D-galactal (CAS 34948-79-3)
QUICK LINKS
Tri-O-benzoyl-D-galactal, a derivative of D-galactal, has emerged as a pivotal reagent in carbohydrate chemistry research, primarily due to its significance in glycosylation reactions and the synthesis of complex carbohydrate structures. The tri-O-benzoyl protection strategy offers chemists precise control over regioselectivity and stereoselectivity during glycosylation processes, allowing for the construction of elaborate oligosaccharides and glycoconjugates with high efficiency and accuracy. This compound serves as a versatile building block in the synthesis of carbohydrate-based materials, bioactive compounds, and natural product analogs, facilitating the exploration of carbohydrate-protein interactions and biological recognition events. Furthermore, Tri-O-benzoyl-D-galactal has been instrumental in the development of novel glycosylation methodologies and the elucidation of carbohydrate biosynthesis pathways. Its utilization spans various fields of research, including chemical biology, medicinal chemistry, and materials science, where it contributes to advancing our understanding of carbohydrate structure-function relationships and the design of carbohydrate-based biomaterials and research tools. Overall, Tri-O-benzoyl-D-galactal stands as a cornerstone in carbohydrate chemistry, enabling groundbreaking research and innovation in diverse scientific disciplines.
Tri-O-benzoyl-D-galactal (CAS 34948-79-3) References
- Glycopeptide specificity of helper T cells obtained in mouse models for rheumatoid arthritis. | Holm, B., et al. 2002. Chembiochem. 3: 1209-22. PMID: 12465029
- Copper mediated stereoselective synthesis of C-glycosides from unactivated alkynes. | Kusunuru, AK., et al. 2013. Chem Commun (Camb). 49: 10154-6. PMID: 24049771
- TMSOTf mediated stereoselective synthesis of α-C-glycosides from unactivated aryl acetylenes. | Chen, H., et al. 2017. Glycoconj J. 34: 13-20. PMID: 27566624
- Advances in C-alkynylation of sugars and its application in organic synthesis[J]. | Tatina M B, Hussain A, Dhas A K,., 2016. RSC advances., 6(79).: 75960-75972.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tri-O-benzoyl-D-galactal, 5 g | sc-255681 | 5 g | $294.00 |
