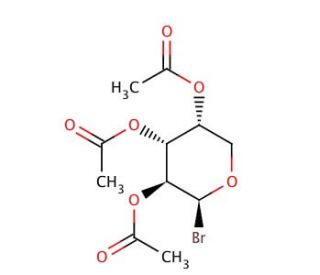
Tri-O-acetyl-β-D-arabinosylbromide (CAS 3068-29-9)
QUICK LINKS
Tri-O-acetyl-β-D-arabinosylbromide has been extensively utilized in scientific research, particularly in the synthesis of arabinosides and related carbohydrate derivatives. As a brominated arabinosyl donor, this compound serves as a key intermediate in the preparation of arabinofuranosides through glycosylation reactions. The mechanism of action involves the activation of the anomeric carbon of the arabinose moiety by the bromide leaving group, facilitating its reaction with an acceptor molecule under appropriate conditions. This chemical has been employed in the synthesis of diverse arabinofuranoside derivatives, including nucleoside analogs, glycoconjugates, and glycolipids. Researchers have utilized these synthetic compounds as tools for probing biological processes involving arabinose-containing molecules, such as glycosylation events in cell surface interactions, host-pathogen interactions, and carbohydrate metabolism. Moreover, the structural versatility of arabinosides has led to their investigation as potential agents in antimicrobial, antiviral, and anticancer drug development. Additionally, tri-O-acetyl-β-D-arabinosylbromide has found applications in carbohydrate chemistry research aimed at elucidating the stereochemical and regioselective aspects of glycosylation reactions, contributing to the advancement of synthetic methodologies and the understanding of carbohydrate structure-function relationships.
Tri-O-acetyl-β-D-arabinosylbromide (CAS 3068-29-9) References
- Synthesis and antitumor activity of quaternary ellipticine glycosides, a series of novel and highly active antitumor agents. | Honda, T., et al. 1988. J Med Chem. 31: 1295-305. PMID: 3385725
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tri-O-acetyl-β-D-arabinosylbromide, 1 g | sc-237237 | 1 g | $49.00 |
