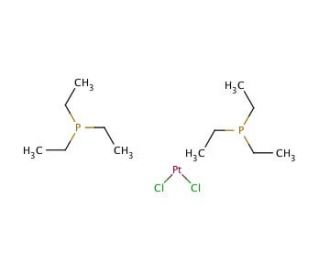
Molecular structure of trans-Dichlorobis(triethylphosphine)platinum(II), CAS Number: 13965-02-1
trans-Dichlorobis(triethylphosphine)platinum(II) (CAS 13965-02-1)
CAS Number:
13965-02-1
Molecular Weight:
502.30
Molecular Formula:
C12H30Cl2P2Pt
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
The structure of trans-Dichlorobis(triethylphosphine)platinum(II) is characterized by the platinum atom at its center, forming a square planar geometry. This geometric arrangement is for the compound′s reactivity and its ability to act as a catalyst in various chemical reactions. The presence of triethylphosphine ligands provides the compound with both steric and electronic effects that influence its catalytic activity and stability. It serves as a nucleophilic reagent, readily engaging in substitution reactions with electron-rich molecules like amines and alcohols. Its versatile nature allows for the synthesis of various chemical compounds through reactions with hydrocarbons or amines.
trans-Dichlorobis(triethylphosphine)platinum(II) (CAS 13965-02-1) References
- A star-shaped ruthenium complex with five ferrocenyl-terminated arms bridged by trans-platinum fragments. | Vives, G., et al. 2006. Chem Commun (Camb). 2283-5. PMID: 16718330
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
trans-Dichlorobis(triethylphosphine)platinum(II), 1 g | sc-229534 | 1 g | $281.00 |
