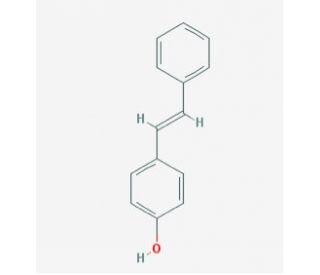
Molecular structure of trans-4-Hydroxystilbene, CAS Number: 6554-98-9
trans-4-Hydroxystilbene (CAS 6554-98-9)
Alternate Names:
p-Hydroxystilbene; p-Styrylphenol
Application:
trans-4-Hydroxystilbene is a phenol compound used for biochemical research
CAS Number:
6554-98-9
Purity:
≥95%
Molecular Weight:
196.24
Molecular Formula:
C14H12O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Trans-4-Hydroxystilbene is a stilbene compound found in various plant species. It is a derivative of resveratrol and is known for its antioxidant properties. In biochemistry, trans-4-Hydroxystilbene has been studied for its potential role in modulating various cellular processes. It exhibits anti-inflammatory effects by inhibiting the expression of pro-inflammatory mediators. Trans-4-Hydroxystilbene has anti-cancer properties, with studies suggesting its ability to induce apoptosis in cancer cells.
trans-4-Hydroxystilbene (CAS 6554-98-9) References
- Metabolic activation of the proestrogens trans-stilbene and trans-stilbene oxide by rat liver microsomes. | Sugihara, K., et al. 2000. Toxicol Appl Pharmacol. 167: 46-54. PMID: 10936078
- Efficiency and mechanism of the antioxidant action of trans-resveratrol and its analogues in the radical liposome oxidation. | Stojanović, S., et al. 2001. Arch Biochem Biophys. 391: 79-89. PMID: 11414688
- Cytochrome P450 1A1/2 mediated metabolism of trans-stilbene in rats and humans. | Sanoh, S., et al. 2002. Biol Pharm Bull. 25: 397-400. PMID: 11913543
- A common structure of substrate shared by lignostilbenedioxygenase isozymes from Sphingomonas paucimobilis TMY1009. | Kamoda, S., et al. 2003. Biosci Biotechnol Biochem. 67: 1394-6. PMID: 12843670
- Possible origin of the skin sensitization potential of isoeugenol and related compounds. (I). Preliminary studies of potential reaction mechanisms. | Barratt, MD. and Basketter, DA. 1992. Contact Dermatitis. 27: 98-104. PMID: 1395636
- Pharmacological inhibition of outwardly rectifying Cl- currents in rat peritoneal mast cells: a comparison of different stilbene derivatives. | Roloff, T., et al. 2003. Eur J Pharmacol. 478: 81-8. PMID: 14575791
- Oxyresveratrol and resveratrol are potent antioxidants and free radical scavengers: effect on nitrosative and oxidative stress derived from microglial cells. | Lorenz, P., et al. 2003. Nitric Oxide. 9: 64-76. PMID: 14623172
- Antihyperuricemic and nephroprotective effects of resveratrol and its analogues in hyperuricemic mice. | Shi, YW., et al. 2012. Mol Nutr Food Res. 56: 1433-44. PMID: 22865646
- The 4'-hydroxyl group of resveratrol is functionally important for direct activation of PPARα. | Takizawa, Y., et al. 2015. PLoS One. 10: e0120865. PMID: 25798826
- Effects of substitution groups of glutamide-derived molecular gels on molecular shape recognition. | Noguchi, H., et al. 2015. J Chromatogr A. 1392: 56-62. PMID: 25817707
- Rapid identification and global characterization of multiple constituents from the essential oil of Cortex Dictamni based on GC-MS. | Zhai, W., et al. 2017. J Sep Sci. 40: 2671-2681. PMID: 28493524
- Antioxidant Activity and Neuroprotective Activity of Stilbenoids in Rat Primary Cortex Neurons via the PI3K/Akt Signalling Pathway. | Wen, H., et al. 2018. Molecules. 23: PMID: 30213108
- Mesoscale Polymer Surfactants: Photolithographic Production and Localization at Droplet Interfaces. | Yang, Z., et al. 2022. J Am Chem Soc. 144: 22059-22066. PMID: 36442142
- Microbial models of mammalian metabolism. Aromatic hydroxylation. | Smith, RV. and Rosazza, JP. 1974. Arch Biochem Biophys. 161: 551-8. PMID: 4209136
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
trans-4-Hydroxystilbene, 5 g | sc-363468 | 5 g | $104.00 |
