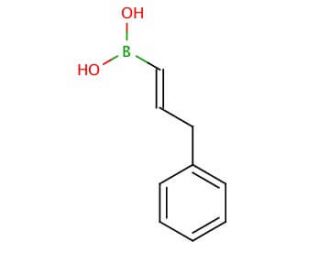
trans-3-Phenyl-1-propen-1-ylboronic acid (CAS 129423-29-6)
QUICK LINKS
Trans-3-Phenylpropen-1-yl-boronic acid is a transparent solid with water solubility and a melting point of 113-114°C. This compound serves multiple purposes in different domains, such as being a fundamental component for organic synthesis, an effective catalyst for various reactions, and a ligand for complexes involving transition metals. Widely employed in scientific research applications, Trans-3-Phenyl-1-propen-1-ylboronic acid is extensively utilized as a building block for organic synthesis, a catalyst for diverse reactions, and a ligand for transition metal complexes. Additionally, it acts as a substrate for enzymes, a reagent for the synthesis of boronates, borates, and boron-containing compounds, and demonstrates electrophilic, nucleophilic, and catalytic properties in various reactions.
trans-3-Phenyl-1-propen-1-ylboronic acid (CAS 129423-29-6) References
- Quinine-Promoted, Enantioselective Boron-Tethered Diels-Alder Reaction by Anomeric Control of Transition-State Conformation. | Scholl, K., et al. 2018. J Org Chem. 83: 5756-5765. PMID: 29715015
- Biomimetic electrochemical sensor for the highly selective detection of azithromycin in biological samples. | Stoian, IA., et al. 2020. Biosens Bioelectron. 155: 112098. PMID: 32090870
- N-Heterocyclic Carbene (NHC)-Rhodium-Catalyzed Carbonylative CC Bond Formation of Allenols with Arylboronic Acids under Carbon Monoxide | Soo Young Choi, Young Keun Chung. 2011. Advanced Synthesis & Catalysis. 353: 2609-2613.
- Facile Preparation of Vinyl S-Trifluoromethyl NH Aryl Sulfoximines | Anne-Laure Barthelemy, Alexis Prieto, Patrick Diter, Jérôme Hannedouche, Martial Toffano, Elsa Anselmi, Emmanuel Magnier. 2018. European Journal of Organic Chemistry. 2018: 3764-3770.
- Direct Electrochemistry and Sensitive Detection of Guanosine on Nanopolymeric Surfaces Bearing Boronic Acid Groups | Prof. Filiz Kuralay, Taner Gürsoy. 2020. ChemistrySelect. 5: 9134-9142.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
trans-3-Phenyl-1-propen-1-ylboronic acid, 1 g | sc-253726 | 1 g | $270.00 |
