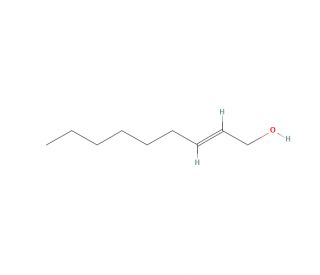
trans-2-Nonen-1-ol (CAS 31502-14-4)
QUICK LINKS
trans-2-Nonen-1-ol is a compound that has applications in research areas such as organic chemistry and materials science, particularly due to its unsaturated alcohol structure. It serves as a starting material or intermediate in the synthesis of various organic compounds, where the presence of both a double bond and a hydroxyl group allows for diverse chemical modifications. Studies involving trans-2-Nonen-1-ol often explore its reactivity in reactions such as catalytic hydrogenation, oxidation, or halogenation. The double bond can undergo addition reactions, which are useful for modifying the compound′s chain length or introducing new functional groups. In the field of flavor and fragrance chemistry, research on trans-2-Nonen-1-ol may focus on its role in the synthesis of aroma compounds, where its structural components contribute to generating complex scent profiles. Additionally, its alcohol functionality provides an anchor for the compound to be used in the preparation of esters and ethers, which can be studied for their potential applications in various industrial processes.
trans-2-Nonen-1-ol (CAS 31502-14-4) References
- Evaluation of the potential effects of ingredients added to cigarettes. Part 1: cigarette design, testing approach, and review of results. | Carmines, EL. 2002. Food Chem Toxicol. 40: 77-91. PMID: 11731038
- The FEMA GRAS assessment of alpha,beta-unsaturated aldehydes and related substances used as flavor ingredients. | Adams, TB., et al. 2008. Food Chem Toxicol. 46: 2935-67. PMID: 18644420
- Volatile composition of Catharanthus roseus (L.) G. Don using solid-phase microextraction and gas chromatography/mass spectrometry. | De Pinho, PG., et al. 2009. J Pharm Biomed Anal. 49: 674-85. PMID: 19186019
- Volatile compounds of healthy and insect-damaged Hippophae rhamnoides sinensis in natural and planted forests. | Zong, S., et al. 2012. Z Naturforsch C J Biosci. 67: 244-8. PMID: 22888528
- Influence of oxygen and long term storage on the profile of volatile compounds released from polymeric multilayer food contact materials sterilized by gamma irradiation. | Salafranca, J., et al. 2015. Anal Chim Acta. 878: 118-30. PMID: 26002333
- Odorant receptor-based discovery of natural repellents of human lice. | Pelletier, J., et al. 2015. Insect Biochem Mol Biol. 66: 103-9. PMID: 26494014
- Rational Design of a Polyoxometalate Intercalated Layered Double Hydroxide: Highly Efficient Catalytic Epoxidation of Allylic Alcohols under Mild and Solvent-Free Conditions. | Li, T., et al. 2017. Chemistry. 23: 1069-1077. PMID: 27748545
- A GC-MS untargeted metabolomics approach for the classification of chemical differences in grape juices based on fungal pathogen. | Schueuermann, C., et al. 2019. Food Chem. 270: 375-384. PMID: 30174061
- A Novel Discovery: Holistic Efficacy at the Special Organ Level of Pungent Flavored Compounds from Pungent Traditional Chinese Medicine. | Chen, Z., et al. 2019. Int J Mol Sci. 20: PMID: 30754631
- Selected Species of the Cucurbitaceae Family Used in Mexico for the Treatment of Diabetes Mellitus. | Huerta-Reyes, M., et al. 2022. Molecules. 27: PMID: 35684376
- Smartphone-Based Image Analysis for Rapid Evaluation of Kiwifruit Quality during Cold Storage. | Li, H., et al. 2022. Foods. 11: PMID: 35885355
- Physicochemical property, bacterial diversity, and volatile profile during ripening of naturally fermented dry mutton sausage produced from Jianzhou big-eared goat. | Chen, J., et al. 2022. Front Microbiol. 13: 961117. PMID: 36118189
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
trans-2-Nonen-1-ol, 10 ml | sc-486915 | 10 ml | $224.00 |
