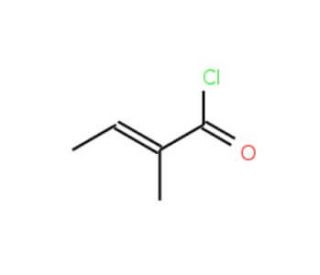
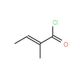
Molecular structure of trans-2-Methyl-2-butenoyl chloride, CAS Number: 35660-94-7
trans-2-Methyl-2-butenoyl chloride (CAS 35660-94-7)
Alternate Names:
Tigloyl chloride; 3-cis-Methyl-methacryloyl chloride; (E)-2-Methylcrotonoyl chloride
CAS Number:
35660-94-7
Molecular Weight:
118.56
Molecular Formula:
C5H7ClO
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
trans-2-Methyl-2-butenoyl chloride possesses the chemical formula C5H7ClO and falls within the category of organochloride compounds. This colorless liquid showcases a potent scent and serves a pivotal role in the creation of diverse organic compounds. Its application extends to catalyzing organic compound synthesis, serving as a reagent for heterocyclic compound formation, and functioning as a fundamental constituent in peptide and protein synthesis, along with the production of fluorescent dyes and other substances.
trans-2-Methyl-2-butenoyl chloride (CAS 35660-94-7) References
- Structure-activity relationship study at the 3'-N-position of paclitaxel: synthesis and biological evaluation of 3'-N-acyl-paclitaxel analogues. | Roh, EJ., et al. 2002. Bioorg Med Chem. 10: 3145-51. PMID: 12150859
- Concise synthesis of anti-HIV-1 active (+)-inophyllum B and (+)-calanolide A by application of (-)-quinine-catalyzed intramolecular oxo-Michael addition. | Sekino, E., et al. 2004. J Org Chem. 69: 2760-7. PMID: 15074925
- Structure reassignment and synthesis of Jenamidines A1/A2, synthesis of (+)-NP25302, and formal synthesis of SB-311009 analogues. | Duvall, JR., et al. 2006. J Org Chem. 71: 8579-90. PMID: 17064037
- Identification of shikonin and its ester derivatives from the roots of Echium italicum L. | Albreht, A., et al. 2009. J Chromatogr A. 1216: 3156-62. PMID: 19233376
- Gibepyrone Biosynthesis in the Rice Pathogen Fusarium fujikuroi Is Facilitated by a Small Polyketide Synthase Gene Cluster. | Janevska, S., et al. 2016. J Biol Chem. 291: 27403-27420. PMID: 27856636
- Therapeutic Evaluation of Synthetic Peucedanocoumarin III in an Animal Model of Parkinson's Disease. | Ham, S., et al. 2019. Int J Mol Sci. 20: PMID: 31689937
- Metabolomics-guided discovery of cytochrome P450s involved in pseudotropine-dependent biosynthesis of modified tropane alkaloids. | Sadre, R., et al. 2022. Nat Commun. 13: 3832. PMID: 35780230
- Synthesis and local anesthetic properties of secondary alkoxyalkylaminoacylanilides. | Kronberg, GH., et al. 1978. J Pharm Sci. 67: 595-600. PMID: 641788
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
trans-2-Methyl-2-butenoyl chloride, 1 g | sc-237165 | 1 g | $97.00 |
