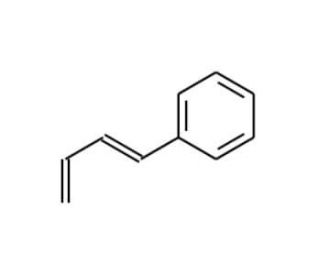
Molecular structure of trans-1-Phenyl-1,3-butadiene, CAS Number: 16939-57-4
trans-1-Phenyl-1,3-butadiene (CAS 16939-57-4)
Alternate Names:
[(E)-1,3-Butadienyl]benzene
Application:
trans-1-Phenyl-1,3-butadiene is an organic building block
CAS Number:
16939-57-4
Purity:
≥95%
Molecular Weight:
130.19
Molecular Formula:
C10H10
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
trans-1-Phenyl-1,3-butadiene is an organic building block. trans-1-Phenyl-1,3-butadiene is an alicyclic hydrocarbon with limited solubility in water. Its versatility makes it a valuable starting material in the synthesis of various organic compounds, including polymers and dyes. Moreover, this compound serves as a reagent in the laboratory for diverse reactions like the Grignard reaction and the Wittig reaction. Additionally, it plays a role in the synthesis of other alicyclic hydrocarbons such as 1-Phenyl-2-butadiene, 1-Phenyl-3-butadiene, and 1-Phenyl-4-butadiene.
trans-1-Phenyl-1,3-butadiene (CAS 16939-57-4) References
- Synthesis of highly functionalized tri- and tetrasubstituted alkenes via Pd-catalyzed 1,2-hydrovinylation of terminal 1,3-dienes. | Saini, V., et al. 2015. J Am Chem Soc. 137: 608-11. PMID: 25555197
- Interception and characterization of catalyst species in rhodium bis(diazaphospholane)-catalyzed hydroformylation of octene, vinyl acetate, allyl cyanide, and 1-phenyl-1,3-butadiene. | Nelsen, ER., et al. 2015. J Am Chem Soc. 137: 14208-19. PMID: 26466510
- A Diverted Aerobic Heck Reaction Enables Selective 1,3-Diene and 1,3,5-Triene Synthesis through C-C Bond Scission. | McAlpine, NJ., et al. 2018. J Am Chem Soc. 140: 13634-13639. PMID: 30289691
- Synthesis of Benzylureas and Related Amine Derivatives via Copper-Catalyzed Three-Component Carboamination of Styrenes. | Kennedy-Ellis, JJ., et al. 2020. Org Lett. 22: 8365-8369. PMID: 33074005
- Vinylazaarenes as dienophiles in Lewis acid-promoted Diels-Alder reactions. | Davis, AE., et al. 2021. Chem Sci. 12: 15947-15952. PMID: 35024118
- Origin of Catalysis and Selectivity in Lewis Acid-Promoted Diels-Alder Reactions Involving Vinylazaarenes as Dienophiles. | Portela, S. and Fernández, I. 2022. J Org Chem. 87: 9307-9315. PMID: 35794859
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
trans-1-Phenyl-1,3-butadiene, 1 g | sc-229478 | 1 g | $189.00 |
