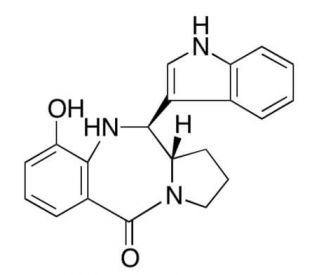
Molecular structure of Tilivalline, CAS Number: 80279-24-9
Tilivalline (CAS 80279-24-9)
See product citations (1)
Application:
Tilivalline is a metabolite of Klebsiella oxytoca, proven to be linked to antibiotic-associated Hemorrhagic Colitis.
CAS Number:
80279-24-9
Molecular Weight:
333.38
Molecular Formula:
C20H19N3O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Tilivalline is a metabolite of Klebsiella oxytoca known to be cytotoxic. The metabolization of Tilivalline is proven to be linked to antibiotic-associated Hemorrhagic Colitis. The derivatives of Tilivalline were able to be synthesized efficiently and stereoselectively from diphenyl phosphorazidate, the 2-oxazoline, L-proline derivatives 5, and Indole in a Mannich-type intramolecular cyclization reaction.
Tilivalline (CAS 80279-24-9) References
- Stereoselective Synthesis of Tilivalline(1). | Nagasaka, T. and Koseki, Y. 1998. J Org Chem. 63: 6797-6801. PMID: 11672297
- Cytotoxic and pathogenic properties of Klebsiella oxytoca isolated from laboratory animals. | Darby, A., et al. 2014. PLoS One. 9: e100542. PMID: 25057966
- A tricyclic pyrrolobenzodiazepine produced by Klebsiella oxytoca is associated with cytotoxicity in antibiotic-associated hemorrhagic colitis. | Tse, H., et al. 2017. J Biol Chem. 292: 19503-19520. PMID: 28972161
- Biosynthesis of the Enterotoxic Pyrrolobenzodiazepine Natural Product Tilivalline. | Dornisch, E., et al. 2017. Angew Chem Int Ed Engl. 56: 14753-14757. PMID: 28977734
- Biosynthesis of the Klebsiella oxytoca Pathogenicity Factor Tilivalline: Heterologous Expression, in Vitro Biosynthesis, and Inhibitor Development. | von Tesmar, A., et al. 2018. ACS Chem Biol. 13: 812-819. PMID: 29389112
- Klebsiella oxytoca enterotoxins tilimycin and tilivalline have distinct host DNA-damaging and microtubule-stabilizing activities. | Unterhauser, K., et al. 2019. Proc Natl Acad Sci U S A. 116: 3774-3783. PMID: 30808763
- Tilivalline- and Tilimycin-Independent Effects of Klebsiella oxytoca on Tight Junction-Mediated Intestinal Barrier Impairment. | Hering, NA., et al. 2019. Int J Mol Sci. 20: PMID: 31717457
- Simultaneous quantification of enterotoxins tilimycin and tilivalline in biological matrices using HPLC high resolution ESMS2 based on isotopically 15N-labeled internal standards. | Glabonjat, RA., et al. 2021. Talanta. 222: 121677. PMID: 33167283
- cAMP Receptor Protein Positively Regulates the Expression of Genes Involved in the Biosynthesis of Klebsiella oxytoca Tilivalline Cytotoxin. | Rodríguez-Valverde, D., et al. 2021. Front Microbiol. 12: 743594. PMID: 34659176
- Klebsiella oxytoca Complex: Update on Taxonomy, Antimicrobial Resistance, and Virulence. | Yang, J., et al. 2022. Clin Microbiol Rev. 35: e0000621. PMID: 34851134
- Bacterial Indole as a Multifunctional Regulator of Klebsiella oxytoca Complex Enterotoxicity. | Ledala, N., et al. 2022. mBio. 13: e0375221. PMID: 35073747
- Wide difference between the cytotoxicity of the 11-alpha-and 11-beta-cyano analogues of tilivalline and their epimeric conversion. | Kohda, K., et al. 1995. Biochem Pharmacol. 49: 1063-8. PMID: 7748186
- New Methods And Reagents In Organic Synthesis. 92. A Stereoselective Synthesis Of Tilivalline And Its Analogs | Shigehiro Mori, et al. 1991. Tetrahedron. 47: 5051-5070.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tilivalline, 1 mg | sc-475699 | 1 mg | $454.00 |
