Tiazofurin (CAS 60084-10-8)
QUICK LINKS
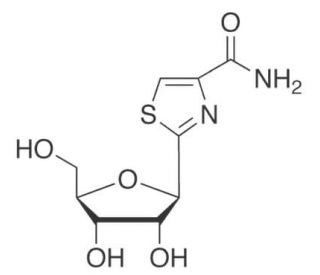
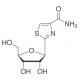
Tiazofurin is a man-made nucleoside analogue that demonstrates antineoplastic activity. Once inside a cell, Tiazofurin is converted to Tiazole-4-carboxamide adenine dinucleotide (TAD), an analogue of NAD. This compound is a potent inhibitor of IMP dehydrogenase (IMPDH), an enzyme that plays a role in purine synthesis. By inhibiting IMPDH, TAD decreases the levels of guanylates, thereby impeding cell growth. Tiazofurin is a C-glycosyl compound originating from beta-D-ribose and is a member of 1,3-thiazoles and monocarboxylic acid amides. As a prodrug, it metabolizes into TAD, which selectively inhibits inosine monophosphate dehydrogenase. Given its role as a potential inhibitor of Inosine- 5′-monophosphate (IMP) dehydrogenase, Tiazofurin has potential utility in certain scientific applications.
Tiazofurin (CAS 60084-10-8) References
- Antitumor activity of tiazofurin in human colon carcinoma HT-29. | Zhen, W., et al. 1992. Cancer Invest. 10: 505-11. PMID: 1358409
- Nicotinamide mononucleotide adenylyltransferase2 overexpression enhances colorectal cancer cell-kill by Tiazofurin. | Kusumanchi, P., et al. 2013. Cancer Gene Ther. 20: 403-12. PMID: 23764899
- Unique bioactivation of tiazofurin--studies with resistant cells. | Kuttan, R. 1989. Indian J Biochem Biophys. 26: 160-5. PMID: 2620910
- Mechanism of resistance to tiazofurin in hepatoma 3924A. | Jayaram, HN., et al. 1986. Biochem Pharmacol. 35: 587-93. PMID: 2868729
- Enzyme-pattern-targeted chemotherapy with tiazofurin and allopurinol in human leukemia. | Weber, G., et al. 1988. Adv Enzyme Regul. 27: 405-33. PMID: 2907968
- Tiazofurin induction of mouse erythroleukemia cell hemoglobin production in the absence of commitment or changes in protooncogene expression. | Sherman, ML., et al. 1989. Blood. 73: 431-4. PMID: 2917182
- Tiazofurin metabolism in human lymphoblastoid cells: evidence for phosphorylation by adenosine kinase and 5'-nucleotidase. | Fridland, A., et al. 1986. Cancer Res. 46: 532-7. PMID: 3000575
- Therapeutic synergism of tiazofurin and selected antitumor drugs against sensitive and resistant P388 leukemia in mice. | Harrison, SD., et al. 1986. Cancer Res. 46: 3396-400. PMID: 3708573
- Selective sensitivity to tiazofurin of human leukemic cells. | Jayaram, HN., et al. 1986. Biochem Pharmacol. 35: 2029-32. PMID: 3718544
- Biochemical consequences of resistance to tiazofurin in human myelogenous leukemic K562 cells. | Jayaram, HN., et al. 1993. Cancer Res. 53: 2344-8. PMID: 8097964
- Tiazofurin enhances the anabolism and toxicity of 5-fluorouracil. | Cysyk, RL., et al. 1996. Cancer Lett. 109: 49-55. PMID: 9020902
- Tiazofurin-induced autosecretion of IL-6 and hemoglobin production in K562 human leukemia cells. | Talley, CJ., et al. 1997. Am J Hematol. 54: 301-5. PMID: 9092685
- Synergistic action of tiazofurin and genistein in human ovarian carcinoma cells. | Li, W. and Weber, G. 1998. Oncol Res. 10: 117-22. PMID: 9700722
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tiazofurin, 5 mg | sc-475805 | 5 mg | $449.00 |
