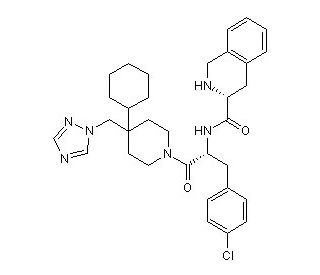
Molecular structure of THIQ, CAS Number: 312637-48-2
THIQ (CAS 312637-48-2)
See product citations (1)
Alternate Names:
N-[(1R)-1-[(4-Chlorophenyl)methyl]-2-[4-cyclohexyl-4-(1H-1,2,4-trazol-1-ylmethyl)-1-piperidinyl]-2-oxoethyl]-1,2,3,4-tetrahydro-3-isoquinolinecarboxamide
Application:
THIQ is a selective melanocortin 4 receptor agonist
CAS Number:
312637-48-2
Purity:
>95%
Molecular Weight:
589.17
Molecular Formula:
C33H41ClN6O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Selective and potent melanocortin 4 (MC4) receptor agonist (IC50 values are 1.2, 761 and 2067 nM for human MC4, MC3 and MC1 receptors respectively). Following systemic administration, enhances intracavernosal pressure and stimulates erectile activity in rats ex copula.
THIQ (CAS 312637-48-2) References
- Functional evaluation of THIQ, a melanocortin 4 receptor agonist, in models of food intake and inflammation. | Muceniece, R., et al. 2007. Basic Clin Pharmacol Toxicol. 101: 416-20. PMID: 18028105
- Key amino acid residues in the melanocortin-4 receptor for nonpeptide THIQ specific binding and signaling. | Yang, Y., et al. 2009. Regul Pept. 155: 46-54. PMID: 19303903
- Discovery of tetrahydroisoquinoline (THIQ) derivatives as potent and orally bioavailable LFA-1/ICAM-1 antagonists. | Zhong, M., et al. 2010. Bioorg Med Chem Lett. 20: 5269-73. PMID: 20655213
- Structure-activity relationship (SAR) of the α-amino acid residue of potent tetrahydroisoquinoline (THIQ)-derived LFA-1/ICAM-1 antagonists. | Zhong, M., et al. 2011. Bioorg Med Chem Lett. 21: 307-10. PMID: 21109434
- Mild-condition synthesis of allenes from alkynes and aldehydes mediated by tetrahydroisoquinoline (THIQ). | Jiang, GJ., et al. 2013. J Org Chem. 78: 11783-93. PMID: 24266693
- A small molecule agonist THIQ as a novel pharmacoperone for intracellularly retained melanocortin-4 receptor mutants. | Huang, H. and Tao, YX. 2014. Int J Biol Sci. 10: 817-24. PMID: 25076858
- An Efficient Nickel-Catalyzed Asymmetric Oxazole-Forming Ugi-Type Reaction for the Synthesis of Chiral Aryl-Substituted THIQ Rings. | Li, D., et al. 2017. Chemistry. 23: 6974-6978. PMID: 28470882
- A novel tetrahydroisoquinoline (THIQ) analogue induces mitochondria-dependent apoptosis. | Sun, X., et al. 2018. Eur J Med Chem. 150: 719-728. PMID: 29573707
- MOF-253-Supported Ru Complex for Photocatalytic CO2 Reduction by Coupling with Semidehydrogenation of 1,2,3,4-Tetrahydroisoquinoline (THIQ). | Deng, X., et al. 2019. Inorg Chem. 58: 16574-16580. PMID: 31774657
- A yeast platform for high-level synthesis of tetrahydroisoquinoline alkaloids. | Pyne, ME., et al. 2020. Nat Commun. 11: 3337. PMID: 32620756
- Conformational and steric aspects of the inhibition of phenylethanolamine N-methyltransferase by benzylamines. | Grunewald, GL., et al. 1988. J Med Chem. 31: 433-44. PMID: 3339613
- 1,2,3,4-Tetrahydroisoquinoline (THIQ) as privileged scaffold for anticancer de novo drug design. | Faheem, ., et al. 2021. Expert Opin Drug Discov. 16: 1119-1147. PMID: 33908322
Inhibitor of:
POMC.Activator of:
BC030500, CKAP4, and MC4-R.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
THIQ, 1 mg | sc-204343 | 1 mg | $260.00 | |||
THIQ, 10 mg | sc-204343A | 10 mg | $1200.00 |
