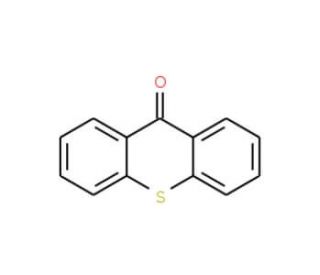
Thioxanthone (CAS 492-22-8)
QUICK LINKS
Thioxanthone, a derivative of xanthone, holds a significant role in various scientific research domains, particularly in the realms of photochemistry and biochemistry. Within the realm of photochemistry, it serves as a valuable photosensitizer, facilitating photochemical reactions like the photobleaching of dyes. In the field of biochemistry, it finds use as an enzyme inhibitor and a fluorescent marker for proteins. Furthermore, in materials science, it serves as a precursor for polymer synthesis and a dopant for semiconductor materials. Thioxanthone′s notable property lies in its capacity to act as a photosensitizer, absorbing light energy and subsequently transferring it to a specific target molecule. This transfer initiates a chemical reaction that has the potential to give rise to the formation of a new molecule.
Thioxanthone (CAS 492-22-8) References
- Thioxanthone: on the shape of the first absorption band. | Rai-Constapel, V., et al. 2010. Phys Chem Chem Phys. 12: 9320-7. PMID: 20607180
- Endocrine-disrupting effects of thioxanthone photoinitiators. | Reitsma, M., et al. 2013. Toxicol Sci. 132: 64-74. PMID: 23208609
- Thioxanthone hydroquinone-O,O'-diacetic acid: photoinitiator or photostabilizer? | Karasu, F., et al. 2013. J Org Chem. 78: 9161-5. PMID: 23944866
- A chiral thioxanthone as an organocatalyst for enantioselective [2+2] photocycloaddition reactions induced by visible light. | Alonso, R. and Bach, T. 2014. Angew Chem Int Ed Engl. 53: 4368-71. PMID: 24648167
- Thioxanthone in apolar solvents: ultrafast internal conversion precedes fast intersystem crossing. | Mundt, R., et al. 2016. Phys Chem Chem Phys. 18: 6637-47. PMID: 26868771
- Structure-Performance Investigation of Thioxanthone Derivatives for Developing Color Tunable Highly Efficient Thermally Activated Delayed Fluorescence Emitters. | Wang, Z., et al. 2016. ACS Appl Mater Interfaces. 8: 8627-36. PMID: 27003610
- Spectrophotometric study on binding of 2-thioxanthone acetic acid with ct-DNA. | Ataci, N., et al. 2018. Spectrochim Acta A Mol Biomol Spectrosc. 204: 281-286. PMID: 29945110
- Silicone-Thioxanthone: A Multifunctionalized Visible Light Photoinitiator with an Ability to Modify the Cured Polymers. | Wu, Q., et al. 2019. Polymers (Basel). 11: PMID: 30995770
- Cytotoxic effects of thioxanthone derivatives as photoinitiators on isolated rat hepatocytes. | Nakagawa, Y., et al. 2020. J Appl Toxicol. 40: 234-244. PMID: 31633820
- A Thioxanthone Sensitizer with a Chiral Phosphoric Acid Binding Site: Properties and Applications in Visible Light-Mediated Cycloadditions. | Pecho, F., et al. 2020. Chemistry. 26: 5190-5194. PMID: 32065432
- Thioxanthone: a powerful photocatalyst for organic reactions. | Nikitas, NF., et al. 2021. Org Biomol Chem. 19: 5237-5253. PMID: 34047729
- Discovery of a Thioxanthone-TfOH Complex as a Photoredox Catalyst for Hydrogenation of Alkenes Using p-Xylene as both Electron and Hydrogen Sources. | Kang, WJ., et al. 2022. Angew Chem Int Ed Engl. 61: e202211562. PMID: 36107463
- Unraveling the Efficiency of Thioxanthone Based Triplet Sensitizers: A Detailed Theoretical Study. | Chandra Garain, B. and Pati, SK. 2023. Chemphyschem. 24: e202200753. PMID: 36495016
- Effect of Substituents with the Different Electron-Donating Abilities on Optoelectronic Properties of Bipolar Thioxanthone Derivatives. | Macionis, S., et al. 2023. ACS Appl Electron Mater. 5: 2227-2238. PMID: 37124238
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Thioxanthone, 50 g | sc-281170 | 50 g | $130.00 |
