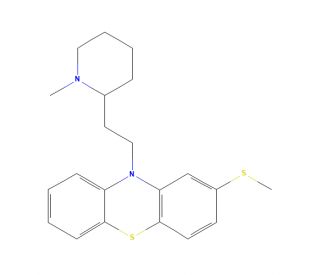
Thioridazine (CAS 50-52-2)
QUICK LINKS
Thioridazine is a compound known for its distinctive chemical structure characterized by a piperidine ring linked to a phenothiazine backbone. This configuration allows it to interact with various molecular targets, making it a useful agent in biochemical research, particularly in the study of cellular signaling pathways and ion channel dynamics. The primary mechanism of action of thioridazine involves its ability to block certain types of ion channels, particularly potassium channels. By modulating the flow of potassium ions through these channels, thioridazine can influence cellular excitability and signaling. This blocking action provides a basis for studying the effects of ion channel modulation on cellular processes such as the firing of neurons and the regulation of muscle contractions. Thioridazine has been utilized to explore the biophysical properties of ion channels and their role in maintaining cellular homeostasis. Its influence on ion channel activity helps researchers understand the underlying mechanisms of ion transport across cell membranes and how these processes affect overall cellular function.
Thioridazine (CAS 50-52-2) References
- Thioridazine treatment prevents cardiopathy in Trypanosoma cruzi infected mice. | Lo Presti, MS., et al. 2004. Int J Antimicrob Agents. 23: 634-6. PMID: 15194137
- The curative activity of thioridazine on mice infected with Mycobacterium tuberculosis. | Martins, M., et al. 2007. In Vivo. 21: 771-5. PMID: 18019410
- Effect of thioridazine on experimental cutaneous staphylococcal infections. | Hahn, BL. and Sohnle, PG. 2014. In Vivo. 28: 33-8. PMID: 24425833
- The Effect of Fluphenazine and Thioridazine on Toxoplasma gondii In Vivo. | Saraei, M., et al. 2016. Iran J Parasitol. 11: 226-231. PMID: 28096857
- Thioridazine inhibits autophagy and sensitizes glioblastoma cells to temozolomide. | Johannessen, TC., et al. 2019. Int J Cancer. 144: 1735-1745. PMID: 30289977
- Thioridazine Induces Cardiotoxicity via Reactive Oxygen Species-Mediated hERG Channel Deficiency and L-Type Calcium Channel Activation. | Liu, Y., et al. 2020. Oxid Med Cell Longev. 2020: 3690123. PMID: 32064022
- Thioridazine aggravates skeletal myositis, systemic and liver inflammation in Trypanosoma cruzi-infected and benznidazole-treated mice. | Mendonça, AAS., et al. 2020. Int Immunopharmacol. 85: 106611. PMID: 32447223
- The combination of cloxacillin, thioridazine and tetracycline protects mice against Staphylococcus aureus peritonitis by inhibiting α-Hemolysin-induced MAPK/NF-κB/NLRP3 activation. | Zhou, H., et al. 2022. Int J Biol Macromol. 198: 1-10. PMID: 34963621
- Repurposing thioridazine for inducing immunogenic cell death in colorectal cancer via eIF2α/ATF4/CHOP and secretory autophagy pathways. | Tran, TH., et al. 2023. Cell Commun Signal. 21: 184. PMID: 37488534
- In Vitro and In Vivo Antileishmanial Activity of Thioridazine. | Sifontes-Rodríguez, S., et al. 2024. Acta Parasitol. 69: 324-331. PMID: 38070122
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Thioridazine, 50 mg | sc-473180 | 50 mg | $500.00 |
