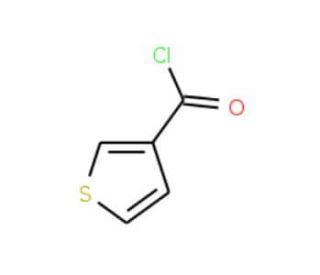
Thiophene-3-carbonyl chloride (CAS 41507-35-1)
QUICK LINKS
Thiophene-3-carbonyl chloride is a reactive compound that functions as an acylating agent in various chemical reactions. It is used in the synthesis of agrochemicals due to its ability to introduce the thiophene-3-carbonyl group into organic molecules. In the presence of suitable nucleophiles, such as amines or alcohols, thiophene-3-carbonyl chloride undergoes acylation to form amides or esters, respectively. Thiophene-3-Carbonyl Chloride′s mechanism of action involves the activation of the carbonyl group, which then reacts with the nucleophile to form the desired acylated product. The resulting compounds can be further modified to create diverse chemical structures for use in development applications. Thiophene-3-carbonyl chloride′s reactivity and selectivity make it useful for chemists seeking to introduce the thiophene-3-carbonyl functionality into organic molecules for experimental purposes.
Thiophene-3-carbonyl chloride (CAS 41507-35-1) References
- Synthesis of bis(ethylenedithio)tetrathiafulvalene (BEDT-TTF) derivatives functionalised with two, four or eight hydroxyl groups. | Brown, RJ., et al. 2007. Org Biomol Chem. 5: 3172-82. PMID: 17878976
- Label-free impedimetric sensor for a ribonucleic acid oligomer specific to hepatitis C virus at a self-assembled monolayer-covered electrode. | Park, JY., et al. 2010. Anal Chem. 82: 8342-8. PMID: 20828200
- The stimulating influence of thienopyrimidine compounds on the adenylyl cyclase signaling systems in the rat testes. | Shpakov, AO., et al. 2014. Dokl Biochem Biophys. 456: 104-7. PMID: 24993967
- [Activation of adenylyl cyclase in rats testes and ovaries using thienopyrimidine derivatives]. | Shpakov, AO., et al. 2014. Tsitologiia. 56: 346-52. PMID: 25696974
- Difunctionalization of Alkenylpyridine N-Oxides by the Tandem Addition/Boekelheide Rearrangement. | Xun, X., et al. 2019. Org Lett. 21: 8266-8269. PMID: 31565945
- Opioid ligands related to tifluadom | Archer, S., Seyed-Mozaffari, A., Simon, E. J., & Gioannini, T. L. 1989. European journal of medicinal chemistry. 24(6): 569-572.
- Bandgap and molecular energy level control of conjugated polymer photovoltaic materials based on benzo [1, 2-b: 4, 5-b′] dithiophene | Hou, J., Park, M. H., Zhang, S., Yao, Y., Chen, L. M., Li, J. H., & Yang, Y. 2008. Macromolecules. 41(16): 6012-6018.
- Low bandgap polymers with benzodithiophene and bisthienylacrylonitrile units for photovoltaic applications | Tan, Z., Imae, I., Ooyama, Y., Komaguchi, K., Ohshita, J., & Harima, Y. 2013. European polymer journal. 49(6): 1634-1641.
- Benzodithiophene‐based poly (aryleneethynylene) s: Synthesis, optical properties, and applications in organic solar cells | Wen, S., Bao, X., Shen, W., Gu, C., Du, Z., Han, L.,.. & Yang, R. 2014. Journal of Polymer Science Part A: Polymer Chemistry. 52(2): 208-215.
- Structural design of benzo [1, 2-b: 4, 5-b′] dithiophene-based 2D conjugated polymers with bithienyl and terthienyl substituents toward photovoltaic applications | Kuo, C. Y., Nie, W., Tsai, H., Yen, H. J., Mohite, A. D., Gupta, G.,.. & Wang, H. L. 2014. Macromolecules. 47(3): 1008-1020.
- C10H4O2S2/graphene composite as a cathode material for sodium-ion batteries | Chen, X., Wu, Y., Huang, Z., Yang, X., Li, W., Yu, L. C.,.. & Chou, S. L. 2016. Journal of Materials Chemistry A. 4(47): 18409-18415.
- Development of a new hybrid CNT-TEPA@ poly (3, 4-ethylenedioxythiophene-co-3-(pyrrol-1-methyl) pyridine) for application as electrode active material in supercapacitors | Lacerda, G. R. D. B. S., dos Santos Junior, G. A., Rocco, M. L. M., Lavall, R. L., Matencio, T., & Calado, H. D. R. 2020. Polymer. 194: 122368.
- Hole-transport materials based on benzodithiophene-thiazolothiazole-containing conjugated polymers for efficient perovskite solar cells | Tepliakova, M. M., Kuznetsov, I. E., Zamoretskov, D. S., Zhivchikova, A. N., Lolaeva, A. V., Furasova, A. D.,.. & Akkuratov, A. V. 2023. Dyes and Pigments. 216: 111349.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Thiophene-3-carbonyl chloride, 1 g | sc-264426 | 1 g | $240.00 | |||
Thiophene-3-carbonyl chloride, 5 g | sc-264426A | 5 g | $459.00 |
