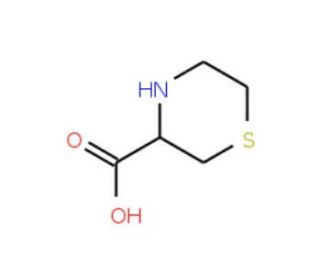
Thiomorpholine-3-carboxylic acid (CAS 20960-92-3)
QUICK LINKS
Thiomorpholine-3-carboxylic acid is a heterocyclic compound that finds application in various research areas within organic chemistry. It is used as a building block in the synthesis of a wide range of sulfur-containing compounds. In the realm of chemical synthesis, thiomorpholine-3-carboxylic acid serves as a key intermediate for the preparation of more complex molecules, particularly those with chiral centers, due to its inherent chirality. Researchers leverage this compound to study the formation of cyclic structures and the introduction of sulfur into peptide-like frameworks. Additionally, it is employed in the development of novel ligands for metal coordination chemistry, where its thiomorpholine moiety can act as a donor atom, forming stable chelates with various metals. This compound is also used to investigate the stability and reactivity of thiomorpholine derivatives under different chemical conditions.
Thiomorpholine-3-carboxylic acid (CAS 20960-92-3) References
- Application of a new chiral derivatizing agent to the enantioseparation of secondary amino acids. | Péter, A., et al. 2002. J Chromatogr A. 948: 283-94. PMID: 12831204
- The reducing activity of S-aminoethylcysteine ketimine and similar sulfur-containing ketimines. | Solinas, SP., et al. 1992. Biochem Biophys Res Commun. 183: 481-6. PMID: 1312831
- Chemical toxicology of reactive intermediates formed by the glutathione-dependent bioactivation of halogen-containing compounds. | Anders, MW. 2008. Chem Res Toxicol. 21: 145-59. PMID: 17696489
- Application of chiral derivatizing agents in the high-performance liquid chromatographic separation of amino acid enantiomers: a review. | Ilisz, I., et al. 2008. J Pharm Biomed Anal. 47: 1-15. PMID: 18242036
- Evaluation of synthetic FK506 analogues as ligands for the FK506-binding proteins 51 and 52. | Gopalakrishnan, R., et al. 2012. J Med Chem. 55: 4114-22. PMID: 22455444
- Enantiomeric separation of nonproteinogenic amino acids by high-performance liquid chromatography. | Ilisz, I., et al. 2012. J Chromatogr A. 1269: 94-121. PMID: 22835690
- Chiral derivatizations applied for the separation of unusual amino acid enantiomers by liquid chromatography and related techniques. | Ilisz, I., et al. 2013. J Chromatogr A. 1296: 119-39. PMID: 23598164
- Identification of 1,4-thiomorpholine-3-carboxylic acid (TMA) in normal human urine. | Matarese, RM., et al. 1989. FEBS Lett. 250: 75-7. PMID: 2737303
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Thiomorpholine-3-carboxylic acid, 250 mg | sc-264423 | 250 mg | $141.00 | |||
Thiomorpholine-3-carboxylic acid, 1 g | sc-264423A | 1 g | $333.00 |
