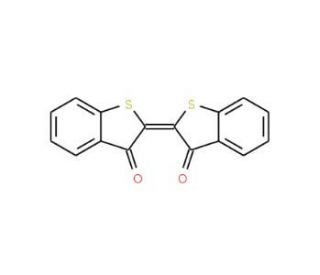
Molecular structure of Thioindigo, CAS Number: 522-75-8
Thioindigo (CAS 522-75-8)
Alternate Names:
Fluorescent Red Dye
CAS Number:
522-75-8
Molecular Weight:
296.36
Molecular Formula:
C16H8O2S2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Thioindigo is an organosulfur compound that belongs to the class of indigo dyes, which are derived from the indigo plant and are used to dye polyester fabric. Thioindigo compounds have been investigated for their potential use in organic electronics and optoelectronic devices, such as organic solar cells, organic light-emitting diodes (OLEDs), and organic field-effect transistors (OFETs). Additionally, the fluorescence and phosphorescence of Thioindigo has led to its use in studies of molecular photophysics and luminescent materials. Furthermore, Thioindigo compounds can serve as versatile building blocks for the synthesis of other complex molecules and materials.
Thioindigo (CAS 522-75-8) References
- Purification, stability, and mineralization of 3-hydroxy-2- formylbenzothiophene, a metabolite of dibenzothiophene. | Bressler, DC. and Fedorak, PM. 2001. Appl Environ Microbiol. 67: 821-6. PMID: 11157249
- Thioindigo dyes: highly accurate visible spectra with TD-DFT. | Jacquemin, D., et al. 2006. J Am Chem Soc. 128: 2072-83. PMID: 16464110
- Organic/inorganic complex pigments: ancient colors Maya Blue. | Polette-Niewold, LA., et al. 2007. J Inorg Biochem. 101: 1958-73. PMID: 17761292
- The Hammett relationship and reactions in the excited electronic state: hemithioindigo Z/E-photoisomerization. | Cordes, T., et al. 2008. J Phys Chem A. 112: 581-8. PMID: 18177026
- Photoacoustic study of the photochemistry of adsorbed indigoid dyes. | Breuer, HD., et al. 1982. Appl Opt. 21: 41-3. PMID: 20372396
- Monothioindigo, determined by microcrystal structure analysis. | Brüning, J., et al. 2010. Acta Crystallogr C. 66: o459-62. PMID: 20814106
- Computational study of the structure of a sepiolite/thioindigo mayan pigment. | Alvarado, M., et al. 2012. Bioinorg Chem Appl. 2012: 672562. PMID: 23193386
- On the spectroscopic analyses of thioindigo dye. | Ibrahim, M., et al. 2013. Spectrochim Acta A Mol Biomol Spectrosc. 113: 332-6. PMID: 23743038
- Synthesis and photophysics of thioindigo diimines and related compounds. | Boice, G., et al. 2014. J Org Chem. 79: 9196-205. PMID: 25198938
- Py-GC/MS applied to the analysis of synthetic organic pigments: characterization and identification in paint samples. | Ghelardi, E., et al. 2015. Anal Bioanal Chem. 407: 1415-31. PMID: 25542568
- Coordination Complexes of Titanium(IV) and Indium(III) Phthalocyanines with Carbonyl-Containing Dyes: The Formation of Singly Bonded Anionic Squarylium Dimers. | Konarev, DV., et al. 2018. Chemistry. 24: 8415-8423. PMID: 29656402
- Mononuclear and Dinuclear Ruthenium Complexes of cis- and trans-Thioindigo: Geometrical and Electronic Structure Analyses. | Chatterjee, M., et al. 2018. Inorg Chem. 57: 12187-12194. PMID: 30198711
- Strong magnetic coupling of spins in Fe(ii) dimers with differently charged thioindigo ligands. | Konarev, DV., et al. 2020. Dalton Trans. 49: 7692-7696. PMID: 32495783
- Ultrafast Excited State Dynamics of Forward and Reverse trans-cis Photoisomerization of Red-Light-Absorbing Indigo Derivatives. | Kihara, Y., et al. 2022. J Phys Chem B. 126: 3539-3550. PMID: 35512335
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Thioindigo, 25 g | sc-296538 | 25 g | $112.00 |
