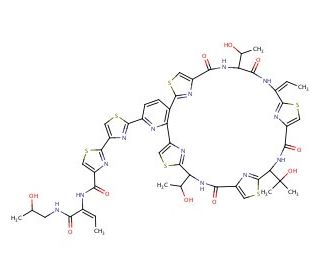
Thiocillin I (CAS 59979-01-0)
QUICK LINKS
Thiocillin I, a thiopeptide antibiotic, is primarily recognized for its role in inhibiting bacterial protein synthesis through a unique mechanism of action that targets the ribosome. Specifically, it binds to the 50S ribosomal subunit, interfering with the assembly of the peptide transferase center, which is crucial for protein translation. This action disrupts the synthesis of essential proteins required for bacterial growth and viability, making thiocillin I a valuable tool in microbiological research aimed at understanding complex ribosomal functions and antibiotic action. Its usage in research extends to studying the mechanisms of action of thiopeptide antibiotics and their roles in combating antibiotic resistance. Thiocillin I is particularly useful in probing the genetic bases of resistance and susceptibility among various bacterial strains, helping to explain how bacteria evolve to resist such potent inhibitors of protein synthesis. Furthermore, this compound assists in the exploration of new antibacterial strategies and the potential modification of existing antibiotics to enhance their efficacy against resistant strains. Research involving thiocillin I continues to provide significant insights into the intricacies of bacterial protein synthesis and the development of resistance, crucial for advancing fundamental microbiology and antibiotic research.
Thiocillin I (CAS 59979-01-0) References
- Thiopeptide biosynthesis featuring ribosomally synthesized precursor peptides and conserved posttranslational modifications. | Liao, R., et al. 2009. Chem Biol. 16: 141-7. PMID: 19246004
- Proteasome inhibitory activity of thiazole antibiotics. | Pandit, B., et al. 2011. Cancer Biol Ther. 11: 43-7. PMID: 21119308
- Total synthesis and complete structural assignment of thiocillin I. | Aulakh, VS. and Ciufolini, MA. 2011. J Am Chem Soc. 133: 5900-4. PMID: 21446660
- Saturation mutagenesis of TsrA Ala4 unveils a highly mutable residue of thiostrepton A. | Zhang, F. and Kelly, WL. 2015. ACS Chem Biol. 10: 998-1009. PMID: 25572285
- The structure of SpnF, a standalone enzyme that catalyzes [4 + 2] cycloaddition. | Fage, CD., et al. 2015. Nat Chem Biol. 11: 256-8. PMID: 25730549
- Total synthesis of micrococcin P1 and thiocillin I enabled by Mo(vi) catalyst. | Akasapu, S., et al. 2019. Chem Sci. 10: 1971-1975. PMID: 30881626
- Virtual Screening Identifies Chebulagic Acid as an Inhibitor of the M2(S31N) Viral Ion Channel and Influenza A Virus. | Duncan, MC., et al. 2020. Molecules. 25: PMID: 32599753
- A Strong Synergy Between the Thiopeptide Bacteriocin Micrococcin P1 and Rifampicin Against MRSA in a Murine Skin Infection Model. | Ovchinnikov, KV., et al. 2021. Front Immunol. 12: 676534. PMID: 34276663
- Dynamic interspecies interactions and robustness in a four-species model biofilm. | Baliarda, A., et al. 2021. Microbiologyopen. 10: e1254. PMID: 34964290
- Catalytic Amide Activation with Thermally Stable Molybdenum(VI) Dioxide Complexes. | Evenson, GE., et al. 2023. J Org Chem. 88: 6192-6202. PMID: 37027833
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Thiocillin I, 500 µg | sc-391644 | 500 µg | $250.00 |
