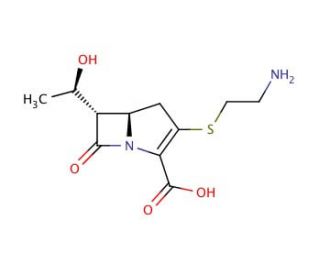
Thienamycin (CAS 59995-64-1)
QUICK LINKS
Thienamycin is a naturally derived compound from the bacterium Streptomyces cattleya, primarily recognized for its potent mechanism of action, which serves as a model for studying bacterial resistance and enzyme inhibition. It plays a crucial role as a precursor in synthesizing various synthetic agents, especially within the carbapenem category, used for research on bacterial cell wall synthesis mechanisms. The primary action of thienamycin involves the inhibition of penicillin-binding proteins (PBPs), which are key to the cross-linking process that provides structural integrity to bacterial cell walls. By binding to these proteins, thienamycin disrupts this critical process, leading to the breakdown of the cell wall structure under natural conditions. In scientific research, thienamycin is extensively used to study the molecular dynamics of PBPs and their role in bacterial growth and survival. Investigating how thienamycin interacts with these proteins provides valuable insights into the structural changes during the interaction. This research aids in understanding how modifications in structure can influence the interactions between such compounds and their targets. Additionally, studying thienamycin helps explore new ways to address bacterial resistance mechanisms, contributing to the broader field of microbiology and enhancing our understanding of bacterial behavior in response to specific molecular interventions.
Thienamycin (CAS 59995-64-1) References
- Identification of transcriptional activators for thienamycin and cephamycin C biosynthetic genes within the thienamycin gene cluster from Streptomyces cattleya. | Rodríguez, M., et al. 2008. Mol Microbiol. 69: 633-45. PMID: 19138192
- Mutational analysis of the thienamycin biosynthetic gene cluster from Streptomyces cattleya. | Rodríguez, M., et al. 2011. Antimicrob Agents Chemother. 55: 1638-49. PMID: 21263049
- Consecutive radical S-adenosylmethionine methylations form the ethyl side chain in thienamycin biosynthesis. | Marous, DR., et al. 2015. Proc Natl Acad Sci U S A. 112: 10354-8. PMID: 26240322
- Formal synthesis of Thienamycin. | Pieczykolan, M., et al. 2017. J Antibiot (Tokyo). 70: 781-787. PMID: 28377636
- Kinetic study on the inhibition of type Ib penicillinase by imipenem and thienamycin. | Hashizume, T., et al. 1988. Chem Pharm Bull (Tokyo). 36: 676-84. PMID: 3044627
- Binding of thienamycin and clavulanic acid to the penicillin-binding proteins of Escherichia coli K-12. | Spratt, BG., et al. 1977. Antimicrob Agents Chemother. 12: 406-9. PMID: 334066
- A metallo-β-lactamase enzyme for internal detoxification of the antibiotic thienamycin. | Diene, SM., et al. 2021. Sci Rep. 11: 10062. PMID: 33980996
- A comparative in vitro study of thienamycin. | Fainstein, V., et al. 1982. Infection. 10: 50-2. PMID: 7068233
- In vitro activity of thienamycin. | Tally, FP., et al. 1978. Antimicrob Agents Chemother. 14: 436-8. PMID: 708019
- In vitro activity of thienamycin. | Shah, PM. 1981. Methods Find Exp Clin Pharmacol. 3: 387-90. PMID: 7329164
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Thienamycin, 1 mg | sc-474865 | 1 mg | $607.00 |
