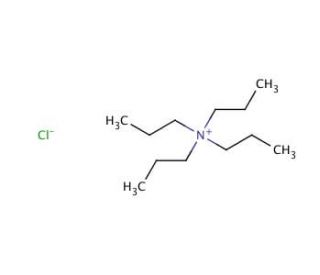
Tetrapropylammonium hydroxide solution (CAS 4499-86-9)
QUICK LINKS
Tetrapropylammonium hydroxide solution (TPAOH) is a widely-used inorganic compound in diverse scientific research applications. Being a strong base with a pKa of 10.5, it readily dissolves in water, alcohols, and other organic solvents. This quaternary ammonium hydroxide finds application in various fields, including organic compound synthesis, metal oxide preparation, and nanoparticle synthesis. Tetrapropylammonium hydroxide solution′s role as a robust base makes it a versatile catalyst for numerous chemical reactions. It displays its versatility as a nucleophile, a Lewis base, and a Bronsted base. Moreover, it facilitates the formation of carbon-carbon bonds and hydrogen bonds, making it a valuable catalyst in those processes.
Tetrapropylammonium hydroxide solution (CAS 4499-86-9) References
- Designing a capsule catalyst and its application for direct synthesis of middle isoparaffins. | He, J., et al. 2005. Langmuir. 21: 1699-702. PMID: 15723460
- Synthesis and characterization of silicalite-1/carbon-graphite membranes. | Titiloye, JO. and Hussain, I. 2008. J Colloid Interface Sci. 318: 50-8. PMID: 17988676
- Characterization of nanoparticles in diluted clear solutions for Silicalite-1 zeolite synthesis using liquid 29Si NMR, SAXS and DLS. | Follens, LR., et al. 2008. Phys Chem Chem Phys. 10: 5574-83. PMID: 18956092
- Rhodamine B immobilized on hollow Au-HMS material for naked-eye detection of Hg2+ in aqueous media. | Zhang, N., et al. 2012. J Hazard Mater. 229-230: 404-10. PMID: 22771346
- The aqueous colloidal suspension of ultrathin 2D MCM-22P crystallites. | Maluangnont, T., et al. 2014. Chem Commun (Camb). 50: 7378-81. PMID: 24872251
- Oxidation of bioethanol using zeolite-encapsulated gold nanoparticles. | Mielby, J., et al. 2014. Angew Chem Int Ed Engl. 53: 12513-6. PMID: 25196739
- Reactivity of fecapentaene-12 toward thiols, DNA, and these constituents in human fibroblasts. | Dypbukt, JM., et al. 1989. Cancer Res. 49: 6058-63. PMID: 2790820
- Characterization at the Level of Individual Crystals: Single-Crystal MFI Type Zeolite Grains. | Gruene, T., et al. 2018. Chemistry. 24: 2384-2388. PMID: 29193398
- Comparative Study between Direct and Pseudomorphic Transformation of Rice Husk Ash into MFI-Type Zeolite. | Alyosef, HA., et al. 2017. Molecules. 23: PMID: 29267245
- ATP3 and MTP3: Easily Prepared Stable Perruthenate Salts for Oxidation Applications in Synthesis. | Moore, PW., et al. 2018. Chemistry. 24: 4556-4561. PMID: 29508453
- Hierarchical Shell-Like ZSM-5 with Tunable Porosity Synthesized by using a Dissolution-Recrystallization Approach. | Lin, J., et al. 2018. Chemistry. 24: 14974-14981. PMID: 30073705
- Ga-Doped and Pt-Loaded Porous TiO2-SiO2 for Photocatalytic Nonoxidative Coupling of Methane. | Wu, S., et al. 2019. J Am Chem Soc. 141: 6592-6600. PMID: 30924338
- Variation of Aluminium Distribution in Small-Sized ZSM-5 Crystals during Desilication. | Li, T., et al. 2019. Chemistry. 25: 15879-15886. PMID: 31553090
- In Situ Recrystallization of Mesoporous Carbon-Silica Composite for the Synthesis of Hierarchically Porous Zeolites. | Du, J., et al. 2020. Materials (Basel). 13: PMID: 32252258
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetrapropylammonium hydroxide solution, 25 g | sc-251215 | 25 g | $35.00 |
