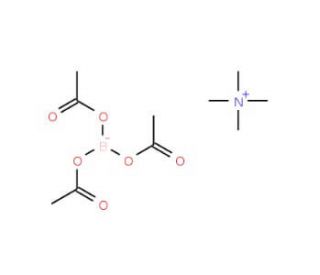
Tetramethylammonium triacetoxyborohydride (CAS 109704-53-2)
QUICK LINKS
Tetramethylammonium triacetoxyborohydride (TMAB) is a potent organic compound extensively utilized in the realm of synthetic organic chemistry. It serves as a robust reducing agent, predominantly employed for converting aldehydes and ketones into their respective alcohols. Beyond this role, Tetramethylammonium triacetoxyborohydride plays a pivotal part in the construction of diverse organic molecules like amines, esters, and amides. Within synthetic organic chemistry, the widespread implementation of Tetramethylammonium triacetoxyborohydride is conspicuous. Its deployment encompasses the creation of a myriad of organic entities, ranging from amines, esters, and amides to polymers, dyes. The capacity of Tetramethylammonium triacetoxyborohydride as a potent reducing agent is unmistakable, particularly in its ability to transform aldehydes and ketones into their corresponding alcohols. The underlying mechanism of this transformation involves the orchestrated transfer of electrons from the boron atom to the carbonyl moiety of the aldehyde or ketone, culminating in the reduction of the carbonyl group to an alcohol.
Tetramethylammonium triacetoxyborohydride (CAS 109704-53-2) References
- Synthetic studies toward (-)-FR901483 using a conjugate allylation to install the C-1 quaternary carbon. | Gotchev, DB. and Comins, DL. 2006. J Org Chem. 71: 9393-402. PMID: 17137366
- Atom-economic and stereoselective syntheses of the ring a and B subunits of the bryostatins. | Trost, BM., et al. 2011. Chemistry. 17: 9777-88. PMID: 21774000
- Development of enantioselective synthetic routes to (-)-kinamycin F and (-)-lomaiviticin aglycon. | Woo, CM., et al. 2012. J Am Chem Soc. 134: 17262-73. PMID: 23030272
- Stereocontrol in Palladium-Catalyzed Propargylic Substitutions: Kinetic Resolution to give Enantioenriched 1,5-Enynes and Propargyl Acetates. | Ardolino, MJ., et al. 2013. Adv Synth Catal. 355: 3413-3419. PMID: 24839434
- Diacyltransferase Activity and Chain Length Specificity of Mycobacterium tuberculosis PapA5 in the Synthesis of Alkyl β-Diol Lipids. | Touchette, MH., et al. 2015. Biochemistry. 54: 5457-68. PMID: 26271001
- Synthesis of a simplified triazole analogue of pateamine A. | Hemi Cumming, A., et al. 2016. Org Biomol Chem. 14: 5117-27. PMID: 27180995
- Identification of novel acetylcholinesterase inhibitors designed by pharmacophore-based virtual screening, molecular docking and bioassay. | Jang, C., et al. 2018. Sci Rep. 8: 14921. PMID: 30297729
- Superior cellular activities of azido- over amino-functionalized ligands for engineered preQ1 riboswitches in E.coli. | Neuner, E., et al. 2018. RNA Biol. 15: 1376-1383. PMID: 30332908
- Total Syntheses of Xiamycins A, C, F, H and Oridamycin A and Preliminary Evaluation of their Anti-Fungal Properties. | Pfaffenbach, M., et al. 2019. Angew Chem Int Ed Engl. 58: 15304-15308. PMID: 31419367
- Facile Synthesis of Saikosaponins. | Wang, Z., et al. 2021. Molecules. 26: PMID: 33808330
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetramethylammonium triacetoxyborohydride, 10 g | sc-258237 | 10 g | $203.00 |
