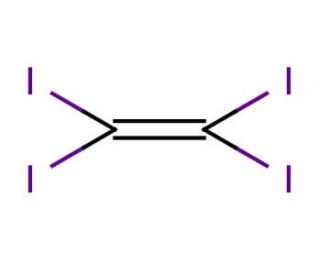
Molecular structure of Tetraiodoethylene, CAS Number: 513-92-8
Tetraiodoethylene (CAS 513-92-8)
See product citations (1)
CAS Number:
513-92-8
Molecular Weight:
531.64
Molecular Formula:
C2I4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Tetraiodoethylene is a synthetic hydrocarbon compound, chemically modified with halogen atoms. The presence of four iodine atoms makes tetraiodoethylene a particularly heavy molecule with substantial steric bulk. It′s used in both scientific and industrial settings. It has found utility in numerous fields, particularly in the synthesis of polymers like polyethylene and polypropylene, contributing to the production of durable and versatile materials. Additionally, it has played a significant role in the creation of dyes and pigments. Tetraiodoethylene can act as a source of iodine in various synthetic chemical reactions, particularly in the synthesis of iodinated organic compounds.
Tetraiodoethylene (CAS 513-92-8) References
- The fungistatic activity of ethylenic and acetylenic compounds; the fungistatic activity of tetraiodoethylene and related compounds. | MUIRHEAD, I. 1949. Ann Appl Biol. 36: 250-6. PMID: 18151951
- Supramolecular insulating networks sheathing conducting nanowires based on organic radical cations. | Yamamoto, HM., et al. 2008. ACS Nano. 2: 143-55. PMID: 19206558
- 1,1-diamino-2,2-dinitroethylenes are always zwitterions. | Gilinsky-Sharon, P., et al. 2012. Magn Reson Chem. 50: 672-9. PMID: 22926948
- The C-I···X¯ halogen bonding of tetraiodoethylene with halide anions in solution and cocrystals investigated by experiment and calculation. | Wang, H., et al. 2013. Phys Chem Chem Phys. 15: 4320-8. PMID: 23407673
- Hexaiododiplatinate(ii) as a useful supramolecular synthon for halogen bond involving crystal engineering. | Eliseeva, AA., et al. 2020. Dalton Trans. 49: 356-367. PMID: 31825414
- Halogen Bonding of Organoiodines and Triiodide Anions in (NMe3 Ph)+ Salts. | Peloquin, AJ., et al. 2021. Chempluschem. 86: 612-621. PMID: 33830671
- Structure-Directing Interplay between Tetrel and Halogen Bonding in Co-Crystal of Lead(II) Diethyldithiocarbamate with Tetraiodoethylene. | Zelenkov, LE., et al. 2022. Int J Mol Sci. 23: PMID: 36233178
- Halogen Bond-Involving Supramolecular Assembly Utilizing Carbon as a Nucleophilic Partner of I⋅⋅⋅C Non-covalent Interaction. | Smirnov, AS., et al. 2023. Chem Asian J. 18: e202300037. PMID: 36807544
- The vibrational spectra and assignments of tetraiodoethylene | EJ Flourie, WD Jones -. March 1969,. Spectrochimica Acta Part A: Molecular Spectroscopy. Volume 25, Issue 3,: Pages 653-659.
- Preparation of polymeric carbon from diiodoacetylene and tetraiodoethylene | and Jaromír Hlavatý, Ladislav Kavan. February 1998. Die Angewandte Makromolekulare Chemie. Volume254, Issue1: Pages 75-78.
- Tetraiodoethylene: a supramolecular host for Lewis base donors | RD Bailey, LL Hook, RP Watson, TW Hanks… -. September 2000,. Crystal Engineering. Volume 3, Issue 3,: Pages 155-171.
- The C–I···–O–N+ Halogen Bonds with Tetraiodoethylene and Aromatic N-Oxides | Khai-Nghi Truong, J. Mikko Rautiainen, Kari Rissanen, and Rakesh Puttreddy*. 2020,. Cryst. Growth Des. 20, 8,: 5330–5337.
- Crystal Structures of a New Polymorph of N-tert-butyl-2-thioimidazole, and Its 1,4-Diiodotetrafluorobenzene, Tetraiodoethylene, and Iodine Cocrystals | , et al. 2022. Journal of Chemical Crystallography volume 52, pages 62–72 (2022)Cite this article. volume 52,: pages 62–72.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetraiodoethylene, 5 g | sc-251192 | 5 g | $247.00 |
