Tetrahydropyran-2-methanol (CAS 100-72-1)
QUICK LINKS
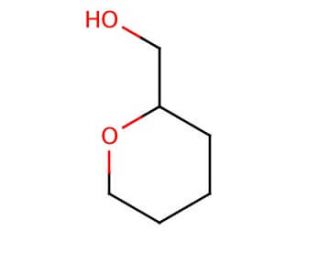
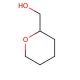
Tetrahydropyran-2-methanol (THPM) is an organic compound classified as a cyclic ether, characterized by its ring structure containing an oxygen atom bound to two carbon atoms. This colorless liquid emits a pungent odor and has found significant use in various scientific research endeavors. In scientific research, Tetrahydropyran-2-methanol serves as a valuable model compound to investigate the reactivity of cyclic ethers, as well as Grignard reagents and other organometallic compounds. Moreover, it plays a role in the synthesis of diverse compounds, including pharmaceuticals and herbicides. Although the exact mechanism of action remains incompletely understood, Tetrahydropyran-2-methanol is believed to interact with specific proteins, enzymes, and receptors within the body to elicit its effects. Depending on the concentration of the compound, it may act as an agonist or antagonist at certain receptors. Tetrahydropyran-2-methanol, a cyclic ether, holds promise in scientific investigations, offering insights into diverse reactivity studies, compound synthesis, and biochemical effects within the body.
Tetrahydropyran-2-methanol (CAS 100-72-1) References
- Structure-activity relationship for adenosine kinase from Mycobacterium tuberculosis II. Modifications to the ribofuranosyl moiety. | Long, MC., et al. 2008. Biochem Pharmacol. 75: 1588-600. PMID: 18329005
- Dominant conformer of tetrahydropyran-2-methanol and its clusters in the gas phase explored by the use of VUV photoionization and vibrational spectroscopy. | Zhan, H., et al. 2017. J Chem Phys. 146: 134303. PMID: 28390350
- Oxidative Amide Coupling from Functionally Diverse Alcohols and Amines Using Aerobic Copper/Nitroxyl Catalysis. | Piszel, PE., et al. 2019. Angew Chem Int Ed Engl. 58: 12211-12215. PMID: 31206988
- Integrated Cascade Process for the Catalytic Conversion of 5-Hydroxymethylfurfural to Furanic and TetrahydrofuranicDiethers as Potential Biofuels. | Fulignati, S., et al. 2022. ChemSusChem. 15: e202200241. PMID: 35384331
- Chemocatalytic value addition of glucose without carbon-carbon bond cleavage/formation reactions: an overview. | Dutta, S. and Bhat, NS. 2022. RSC Adv. 12: 4891-4912. PMID: 35425469
- Rational enzyme design for enabling biocatalytic Baldwin cyclization and asymmetric synthesis of chiral heterocycles. | Li, JK., et al. 2022. Nat Commun. 13: 7813. PMID: 36535947
- Selective inhibition of metabolic enzymes by enzymatically synthesized D-glucal-6-phosphate. | Chenault, HK. and Mandes, RF. 1994. Bioorg Med Chem. 2: 627-9. PMID: 7858968
- Studies on alpha-sialylation using sialyl donors with an auxiliary 3-thiophenyl group. | Martichonok, V. and Whitesides, GM. 1997. Carbohydr Res. 302: 123-9. PMID: 9291567
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetrahydropyran-2-methanol, 5 g | sc-224303 | 5 g | $63.00 |
