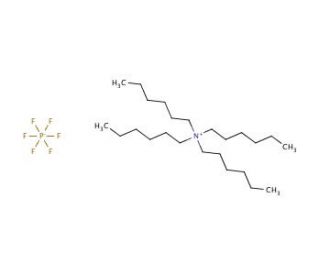
Tetrahexylammonium hexafluorophosphate (CAS 109241-90-9)
QUICK LINKS
Tetrahexylammonium hexafluorophosphate, with its distinct molecular structure comprising a quaternary ammonium ion paired with a hexafluorophosphate counterion, serves as a pivotal reagent in the realm of organic synthesis and materials science research. The unique physicochemical properties of this compound, such as its ionic nature and hydrophobic alkyl chains, endow it with an exceptional ability to act as a phase transfer catalyst. This functionality enables the transfer of reactive anions from aqueous to organic phases, thus facilitating reactions that would otherwise be kinetically unfavorable in a single phase. Moreover, its role is not confined to catalysis alone; tetrahexylammonium hexafluorophosphate is also extensively utilized in the synthesis of novel ionic liquids and as a supporting electrolyte in electrochemical studies. Its contribution to the advancement of materials science is underscored by its application in the development of high-performance polymers and composites, where it is employed to modify surface properties and enhance polymer solubility. Through these diverse roles, tetrahexylammonium hexafluorophosphate exemplifies the critical intersection between innovative chemical design and the pursuit of new technological frontiers in research.
Tetrahexylammonium hexafluorophosphate (CAS 109241-90-9) References
- Voltammetric measurement in supercritical CO2 utilizing a water-in-CO2 microemulsion. | Ohde, H., et al. 2000. Anal Chem. 72: 4738-41. PMID: 11028640
- High-pressure CO2-induced reduction of the melting temperature of ionic liquids. | Kazarian, SG., et al. 2002. Chem Commun (Camb). 1314-5. PMID: 12109131
- Reversible electrochemical generation of a rhodium(II) porphyrin: thwarting disproportionation with weakly coordinating anions. | Sun, H., et al. 2003. Inorg Chem. 42: 4507-9. PMID: 12870935
- Selective silver ion transfer voltammetry at the polarised liquid/liquid interface. | Sherburn, A., et al. 2003. Analyst. 128: 1187-92. PMID: 14529028
- Double-layer effects and distance dependence of electron transfer in reduction of nitro aromatic radical anions. | Morkovská, P., et al. 2006. Langmuir. 22: 1896-902. PMID: 16460125
- Holey gold nanowires formed by photoconversion of dissipative nanostructures emerged at the aqueous-organic interface. | Soejima, T., et al. 2009. Small. 5: 2043-7. PMID: 19462380
- Phase behaviour and conductivity of supporting electrolytes in supercritical difluoromethane and 1,1-difluoroethane. | Han, X., et al. 2016. Phys Chem Chem Phys. 18: 14359-69. PMID: 27166921
- Probing Ion Transfer across Liquid-Liquid Interfaces by Monitoring Collisions of Single Femtoliter Oil Droplets on Ultramicroelectrodes. | Deng, H., et al. 2016. Anal Chem. 88: 7754-61. PMID: 27387789
- Electrochemically Powered, Energy-Conserving Carbon Nanotube Artificial Muscles. | Lee, JA., et al. 2017. Adv Mater. 29: PMID: 28627770
- Recent Advances in Solvents for the Dissolution, Shaping and Derivatization of Cellulose: Quaternary Ammonium Electrolytes and their Solutions in Water and Molecular Solvents. | Kostag, M., et al. 2018. Molecules. 23: PMID: 29495344
- The Role of Onium Salts in the Pro-Oxidant Effect of Gold Nanoparticles in Lipophilic Environments. | Baschieri, A., et al. 2018. Chemistry. 24: 9113-9119. PMID: 29689123
- Continuous and Reversible Tuning of Electrochemical Reaction Kinetics on Back-Gated 2D Semiconductor Electrodes: Steady-State Analysis Using a Hydrodynamic Method. | Kim, CH., et al. 2019. Anal Chem. 91: 1627-1635. PMID: 30569706
- Enhancing the Work Capacity of Electrochemical Artificial Muscles by Coiling Plies of Twist-Released Carbon Nanotube Yarns. | Kim, KJ., et al. 2019. ACS Appl Mater Interfaces. 11: 13533-13537. PMID: 30924629
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetrahexylammonium hexafluorophosphate, 5 g | sc-229417 | 5 g | $225.00 |
