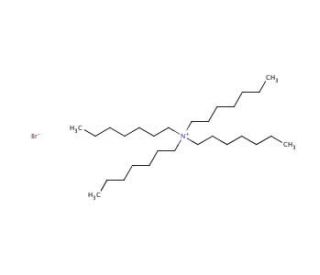
Tetraheptylammonium bromide (CAS 4368-51-8)
QUICK LINKS
Tetraheptylammonium bromide, a quaternary ammonium salt, is notable for its role as a phase-transfer catalyst in chemical synthesis, particularly enhancing the efficiency of reactions involving polar and non-polar phases. This compound consists of a nitrogen atom bonded to four heptyl groups, creating a robust cation, paired with a bromide anion, which collectively facilitates the transfer of reactants across different solvent environments. Its primary mechanism leverages its ionic nature to increase the solubility of polar reactants in non-polar solvents, thus improving reaction kinetics and yields in processes such as nucleophilic substitutions and eliminations. In research settings, tetraheptylammonium bromide is highly valued for its ability to enable more efficient chemical reactions under milder conditions, reducing the need for harsh chemicals and extreme temperatures. This makes it particularly useful in the synthesis of complex organic compounds, including heterocyclic structures, where precise control over reaction conditions is critical. By simplifying and enhancing these synthetic processes, tetraheptylammonium bromide contributes significantly to advancements in organic chemistry, offering a practical solution for overcoming common challenges associated with phase compatibility in reaction mixtures.
Tetraheptylammonium bromide (CAS 4368-51-8) References
- Kinetics of small ion evaporation from the charge and mass distribution of multiply charged clusters in electrosprays. | Gamero-Castano, M. and Mora, JF. 2000. J Mass Spectrom. 35: 790-803. PMID: 10934433
- Kinetics and thermodynamics of hexameric capsule formation. | Yamanaka, M., et al. 2004. J Am Chem Soc. 126: 2939-43. PMID: 14995211
- Development of an analytical method for trace gold in aqueous solution using polyurethane foam sorbents: kinetic and thermodynamic characteristic of gold(III) sorption. | Bashammakh, AS., et al. 2009. Anal Sci. 25: 413-8. PMID: 19276600
- Studies on the formation of hydrophobic ion-pairing complex of alendronate. | You, SK., et al. 2009. Arch Pharm Res. 32: 1055-60. PMID: 19641887
- Solution-processable 2D semiconductors for high-performance large-area electronics. | Lin, Z., et al. 2018. Nature. 562: 254-258. PMID: 30283139
- Retention of Fluorescent Amino Acid Derivatives in Ion-pairing Reversed-phase Liquid Chromatography. | Li, X., et al. 2018. Anal Sci. 34: 1209-1212. PMID: 30305598
- In Situ Probing Molecular Intercalation in Two-Dimensional Layered Semiconductors. | He, Q., et al. 2019. Nano Lett. 19: 6819-6826. PMID: 31498650
- Hydrophobic ion pairing of a GLP-1 analogue for incorporating into lipid nanocarriers designed for oral delivery. | Ismail, R., et al. 2020. Eur J Pharm Biopharm. 152: 10-17. PMID: 32371152
- Exceeding a resolving power of 50 for virus size determination by differential mobility analysis. | Perez-Lorenzo, LJ. and Fernandez de la Mora, J. 2021. J Aerosol Sci. 151: 105658. PMID: 32952209
- Heterogeneous nucleation measurements in a sheathed planar diffusive condensation particle counter. | Kuldinow, D., et al. 2022. J Colloid Interface Sci. 605: 556-570. PMID: 34340040
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetraheptylammonium bromide, 10 g | sc-251183 | 10 g | $101.00 |
