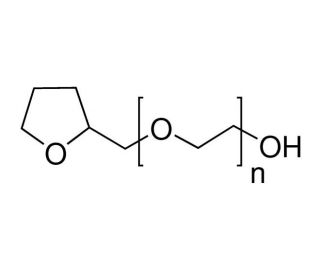
Tetraglycol (CAS 31692-85-0)
QUICK LINKS
Tetraglycol is a chemical compound that functions as a solvent and coupling agent in various experimental applications. It acts by facilitating the dissolution of different substances, thereby promoting the homogeneity of mixtures and enhancing the effectiveness of various processes. At the molecular level, tetraglycol interacts with other compounds through its polar nature, allowing it to form strong intermolecular interactions and promote the dispersion of particles. Tetraglycol can act as a surfactant, reducing the surface tension of liquids and aiding in the emulsification of different substances. Its mechanism of action involves influencing the interfacial properties of solutions, which can be beneficial in various experimental applications.
Tetraglycol (CAS 31692-85-0) References
- Formulation of fast release glibenclamide liquid and semi-solid matrix filled capsules. | Galal, S., et al. 2003. Acta Pharm. 53: 57-64. PMID: 14769253
- Controlled release of paclitaxel from microemulsion containing PLGA and evaluation of anti-tumor activity in vitro and in vivo. | Kang, BK., et al. 2004. Int J Pharm. 286: 147-56. PMID: 15501011
- Rheological, adhesive and release characterisation of semisolid Carbopol/tetraglycol systems. | Bonacucina, G., et al. 2006. Int J Pharm. 307: 129-40. PMID: 16297581
- Acrylic polymers as thickening agents for tetraglycol cosolvent. | Bonacucina, G. and Palmieri, GF. 2006. J Pharm Sci. 95: 726-36. PMID: 16435364
- Effects of solvents and detergents on the contractions of isolated smooth muscle preparations. | Braak, K. and Frey, HH. 1990. J Pharm Pharmacol. 42: 837-41. PMID: 1983145
- Solubilized formulation of olmesartan medoxomil for enhancing oral bioavailability. | Lee, BS., et al. 2009. Arch Pharm Res. 32: 1629-35. PMID: 20091278
- A novel topical targeting system of caffeine microemulsion for inhibiting UVB-induced skin tumor: characterization, optimization, and evaluation. | Ma, H., et al. 2015. AAPS PharmSciTech. 16: 905-13. PMID: 25591953
- Effect of porous polycaprolactone beads on bone regeneration: preliminary in vitro and in vivo studies. | Byun, JH., et al. 2014. Biomater Res. 18: 18. PMID: 26331069
- 3D printing of PLGA scaffolds for tissue engineering. | Mironov, AV., et al. 2017. J Biomed Mater Res A. 105: 104-109. PMID: 27543196
- Supersaturable self-microemulsifying drug delivery system enhances dissolution and bioavailability of telmisartan. | Park, SY., et al. 2021. Pharm Dev Technol. 26: 60-68. PMID: 33032496
- Enhanced oral absorption of insulin: hydrophobic ion pairing and a self-microemulsifying drug delivery system using a D-optimal mixture design. | Goo, YT., et al. 2022. Drug Deliv. 29: 2831-2845. PMID: 36050870
- Isotropic conductive paste for bioresorbable electronics. | Kim, KS., et al. 2023. Mater Today Bio. 18: 100541. PMID: 36647537
- Development of a sorafenib-loaded solid self-nanoemulsifying drug delivery system: Formulation optimization and characterization of enhanced properties. | Lim, C., et al. 2023. J Drug Deliv Sci Technol. 82: PMID: 37124157
- Dimethyl sulfoxide/propylene glycol is a suitable solvent for the delivery of diazepam from osmotic minipumps. | Arnot, MI., et al. 1996. J Pharmacol Toxicol Methods. 36: 29-31. PMID: 8872916
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetraglycol, 100 g | sc-215954 | 100 g | $84.00 | |||
Tetraglycol, 250 g | sc-215954A | 250 g | $124.00 | |||
Tetraglycol, 1 kg | sc-215954B | 1 kg | $349.00 |
