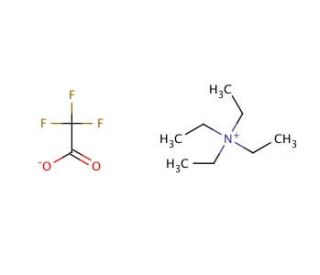
Molecular structure of Tetraethylammonium trifluoroacetate, CAS Number: 30093-29-9
Tetraethylammonium trifluoroacetate (CAS 30093-29-9)
Alternate Names:
Trifluoroacetic acid tetraethylammonium salt
CAS Number:
30093-29-9
Purity:
98%
Molecular Weight:
243.27
Molecular Formula:
C10H20F3NO2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Tetraethylammonium trifluoroacetate (TEA-TFA) is an extensively employed organic compound in scientific research due to its remarkable properties in promoting chemical reactions and enhancing the solubility of specific compounds. This substance serves as an anion-exchange reagent by effectively binding to anions present in a solution. Through this binding, it forms a complex that simplifies the separation of the anion from other constituents within the solution. This separation technique is referred to as anion exchange chromatography.
Tetraethylammonium trifluoroacetate (CAS 30093-29-9) References
- Two new terpyridine dimanganese complexes: a manganese(III,III) complex with a single unsupported oxo bridge and a manganese(III,IV) complex with a dioxo bridge. Synthesis, structure, and redox properties. | Baffert, C., et al. 2002. Inorg Chem. 41: 1404-11. PMID: 11896708
- Quantitative evaluation of the chloride template effect in the formation of dicationic [1(4)]imidazoliophanes. | Ramos, S., et al. 2002. J Org Chem. 67: 8463-8. PMID: 12444626
- Crowned dendrimers: pH-responsive pseudorotaxane formation. | Jones, JW., et al. 2003. J Org Chem. 68: 2385-9. PMID: 12636406
- Remarkably improved complexation of a bisparaquat by formation of a pseudocryptand-based [3]pseudorotaxane. | Huang, F., et al. 2005. Chem Commun (Camb). 1693-5. PMID: 15791301
- Toxicity of ionic liquids: eco(cyto)activity as complicated, but unavoidable parameter for task-specific optimization. | Egorova, KS. and Ananikov, VP. 2014. ChemSusChem. 7: 336-60. PMID: 24399804
- Photo-responsive pseudorotaxanes and assemblies. | Mandal, AK., et al. 2015. Chem Soc Rev. 44: 663-76. PMID: 25438069
- Pseudocryptand Hosts for Paraquats and Diquats. | Jones, JW., et al. 2018. J Org Chem. 83: 823-834. PMID: 29272123
- Renewable Electricity Enables Green Routes to Fine Chemicals and Pharmaceuticals. | Murtaza, A., et al. 2022. Chem Rec. 22: e202100296. PMID: 35103382
- Voltammetric and spectral studies on zirconium in chloride media in acetonitrile | John W. Olver, Russell R. Bessette. 1968. Journal of Inorganic and Nuclear Chemistry. Volume 30, Issue 7: 1791-1798.
- An acid–base adjustable pseudocryptand-type [2]pseudorotaxane based on a bis(meta-phenylene)-32-crown-10 derivative and paraquat | Z Niu, C Slebodnick, F Huang, H Azurmendi. 2011. Tetrahedron Letters. Volume 52, Issue 48: 6379-6382.
- Electrochemical Routes for the Valorization of Biomass-Derived Feedstocks: From Chemistry to Application | Francisco W. S. Lucas, R. Gary Grim, Sean A. Tacey, Courtney A. Downes, Joseph Hasse, Alex M. Roman, Carrie A. Farberow, Joshua A. Schaidle*, and Adam Holewinski*. 2021. ACS Energy Lett. 6, 4: 1205–1270.
- Highly Efficient and Selective Preparation of 5-Hydroxymethylfurfural from Concentrated Carbohydrates Using Deep Eutectic Solvents | Xusheng Guo, Haoxiang Zhu, Yuxi Si, Heng Ban, Xilei Lv, Youwei Cheng*, Lijun Wang, and Xi Li. 2022. ACS Sustainable Chem. Eng. 10, 44: 14579–14587.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetraethylammonium trifluoroacetate, 1 g | sc-251178 | 1 g | $91.00 | |||
Tetraethylammonium trifluoroacetate, 5 g | sc-251178A | 5 g | $406.00 |
