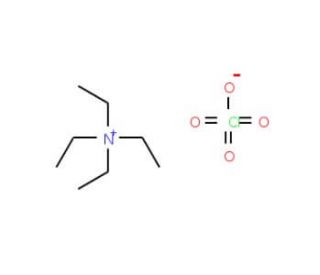
Tetraethylammonium perchlorate (CAS 2567-83-1)
QUICK LINKS
Tetraethylammonium perchlorate (TEAP) holds significant importance as an inorganic compound widely utilized in scientific research. This quaternary ammonium salt consists of a positively charged nitrogen atom bonded to four ethyl groups, making it an organic compound. Tetraethylammonium perchlorate serves diverse scientific research applications. It is employed as a reagent in organic synthesis and as a catalyst in numerous reactions. Moreover, Tetraethylammonium perchlorate plays a vital role in studying the properties of proteins and enzymes. The mechanism of action of Tetraethylammonium perchlorate is intricate, involving various processes. Primarily, it acts as an ionophore, facilitating the transport of ions across membranes.
Tetraethylammonium perchlorate (CAS 2567-83-1) References
- A combined electrochemical quartz-crystal microbalance probe beam deflection (EQCM-PBD) study of solvent and ion transfers at a poly. | Vilas-Boas, M., et al. 2000. Chemistry. 6: 1160-7. PMID: 10785801
- Cyanomethyl anion/carbon dioxide system: an electrogenerated carboxylating reagent. Synthesis of carbamates under mild and safe conditions. | Feroci, M., et al. 2003. J Org Chem. 68: 1548-51. PMID: 12585900
- Hydrolysis of uranium(VI) at variable temperatures (10-85 degrees C). | Zanonato, P., et al. 2004. J Am Chem Soc. 126: 5515-22. PMID: 15113223
- Porous and electrically conductive polypyrrole-poly(vinyl alcohol) composite and its applications as a biomaterial. | Li, Y., et al. 2005. Langmuir. 21: 10702-9. PMID: 16262340
- Solvation effects on the stability of silver(I) complexes with pyridine-containing ligands studied by thermodynamic and DFT methods. | Del Piero, S., et al. 2007. Inorg Chem. 46: 4683-91. PMID: 17480071
- The formation constants of ionomycin with divalent cations in 80% methanol/water. | Stiles, MK., et al. 1991. J Biol Chem. 266: 8336-42. PMID: 1850743
- Development of an analytical method for trace gold in aqueous solution using polyurethane foam sorbents: kinetic and thermodynamic characteristic of gold(III) sorption. | Bashammakh, AS., et al. 2009. Anal Sci. 25: 413-8. PMID: 19276600
- Factors affecting solute entrapment in phospholipid vesicles prepared by the freeze-thaw extrusion method: a possible general method for improving the efficiency of entrapment. | Chapman, CJ., et al. 1990. Chem Phys Lipids. 55: 73-83. PMID: 2090361
- Switching the reaction course of electrochemical CO₂ reduction with ionic liquids. | Sun, L., et al. 2014. Langmuir. 30: 6302-8. PMID: 24851903
- Hydrolysis of thorium(iv) at variable temperatures. | Zanonato, PL., et al. 2016. Dalton Trans. 45: 12763-71. PMID: 27460458
- Molecular Ferroelectric with Most Equivalent Polarization Directions Induced by the Plastic Phase Transition. | Ye, HY., et al. 2016. J Am Chem Soc. 138: 13175-13178. PMID: 27681367
- Ultrafast Polarization Switching in a Biaxial Molecular Ferroelectric Thin Film: [Hdabco]ClO4. | Tang, YY., et al. 2016. J Am Chem Soc. 138: 15784-15789. PMID: 27934003
- [Determination of the by-product 10-bromocarbamazepine in the drug carbamazepine with DC- and differential pulse polarography]. | Dünnbier, U., et al. 1986. Pharmazie. 41: 567-70. PMID: 3786377
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetraethylammonium perchlorate, 10 g | sc-356122 | 10 g | $92.00 | |||
Tetraethylammonium perchlorate, 50 g | sc-356122A | 50 g | $273.00 |
