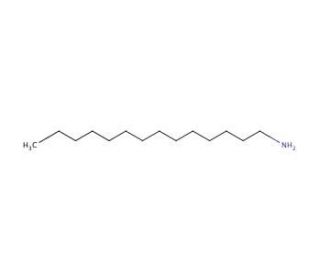
Tetradecylamine (CAS 2016-42-4)
QUICK LINKS
Tetradecylamine is a compound that functions as a surfactant in experimental applications. It acts by reducing the surface tension of liquids, allowing them to spread more easily across solid surfaces. Tetradecylamine′s mechanism of action involves its interaction with the molecules at the interface between the liquid and solid phases, facilitating the wetting and spreading of the liquid. Tetradecylamine achieves this by adsorbing onto the surface, altering the intermolecular forces, and enabling the liquid to form a thin, uniform film. This surfactant′s role is to enhance the wetting and spreading properties of the liquid, which is particularly useful in experimental setups where uniform coverage of a solid surface is required. Its molecular interactions enable it to modify the behavior of the liquid, making it useful for experimental processes that involve surface coatings.
Tetradecylamine (CAS 2016-42-4) References
- [Chromatographic properties of tetradecylamine bonded stationary phase for reversed-phase liquid chromatography]. | Long, Y., et al. 1999. Se Pu. 17: 339-41. PMID: 12552844
- Microbiological activity of certain saturated and unsaturated fatty acid salts of tetradecylamine and related compounds. | BORICK, PM., et al. 1959. Appl Microbiol. 7: 248-51. PMID: 13661870
- A clinical study of the effect of tetradecylamine on plaque formation. | KING, RM. 1951. J Dent Res. 30: 399-402. PMID: 14841317
- Anaerobic degradation of long-chain alkylamines by a denitrifying Pseudomonas stutzeri. | Nguyen, PD., et al. 2008. FEMS Microbiol Ecol. 66: 136-42. PMID: 18721145
- Reaction of alkylamine surfactants with carbon dioxide: relevance to nanocrystal synthesis. | Belman, N., et al. 2009. Nano Lett. 9: 2088-93. PMID: 19374385
- Triggered instability of liposomes bound to hydrophobically modified core-shell PNIPAM hydrogel beads. | MacKinnon, N., et al. 2010. Langmuir. 26: 1081-9. PMID: 19754070
- Surfactant-copper(II) Schiff base complexes: synthesis, structural investigation, DNA interaction, docking studies, and cytotoxic activity. | Lakshmipraba, J., et al. 2015. J Biomol Struct Dyn. 33: 877-91. PMID: 24854148
- Influence of carbon chain length on the synthesis and yield of fatty amine-coated iron-platinum nanoparticles. | Taylor, RM., et al. 2014. Nanoscale Res Lett. 9: 306. PMID: 25006334
- Accurate assessment of the biodegradation of cationic surfactants in activated sludge reactors (OECD TG 303A). | Geerts, R., et al. 2015. Ecotoxicol Environ Saf. 118: 83-89. PMID: 25913361
- Temperature- and pH-Dependent Shattering: Insoluble Fatty Ammonium Phosphate Films at Water-Oil Interfaces. | Forth, J., et al. 2015. Langmuir. 31: 9312-24. PMID: 26263177
- Dodecylamine rapidly kills of spores of multiple Firmicute species: properties of the killed spores and the mechanism of the killing. | DeMarco, AM., et al. 2021. J Appl Microbiol. 131: 2612-2625. PMID: 33998749
- Antimicrobial properties of 2 aliphatic amines and chlorhexidine in vitro and in saliva. | Salem, AM., et al. 1987. J Clin Periodontol. 14: 44-7. PMID: 3543067
- Pickering Emulsions Synergistically Stabilized by Aliphatic Primary Amines and Silica Nanoparticles. | Xue, L., et al. 2022. Langmuir. 38: 14109-14117. PMID: 36349864
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetradecylamine, 5 g | sc-237029 | 5 g | $31.00 |
