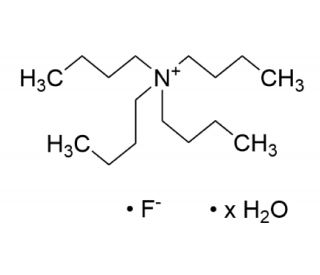
Molecular structure of Tetrabutylammonium Fluoride Hydrate, CAS Number: 22206-57-1
Tetrabutylammonium Fluoride Hydrate (CAS 22206-57-1)
Alternate Names:
TBAF H2O
Application:
Tetrabutylammonium fluoride is a reagent used to prepare terminal olefins from primary alkyl iodides
CAS Number:
22206-57-1
Purity:
98%
Molecular Weight:
261.46 (anhydrous)
Molecular Formula:
C16H36FN•xH2O
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Tetrabutylammonium fluoride (TBAF) is a reagent used to prepare terminal olefins from primary alkyl iodides. Tetrabutylammonium fluoride is also used as a reactant for preparation of double clathrate hydrates at high pressures, cellulose ethers, terminal olefins via dehydrohalogenation reactions, neutral and zwitterionic 3-carboranyl thymidine analogues for boron neutron capture therapy.
Tetrabutylammonium Fluoride Hydrate (CAS 22206-57-1) References
- Anhydrous tetrabutylammonium fluoride. | Sun, H. and DiMagno, SG. 2005. J Am Chem Soc. 127: 2050-1. PMID: 15713075
- Tetrabutylammonium fluoride-assisted rapid N9-alkylation on purine ring: application to combinatorial reactions in microtiter plates for the discovery of potent sulfotransferase inhibitors in situ. | Brik, A., et al. 2005. Bioorg Med Chem. 13: 4622-6. PMID: 15953729
- Tetrabutylammonium fluoride (TBAF)-catalyzed addition of substituted trialkylsilylalkynes to aldehydes, ketones, and trifluoromethyl ketones. | Chintareddy, VR., et al. 2011. J Org Chem. 76: 4482-8. PMID: 21517057
- Simple and convenient synthesis of esters from carboxylic acids and alkyl halides using tetrabutylammonium fluoride. | Matsumoto, K., et al. 2014. J Oleo Sci. 63: 539-44. PMID: 24770480
- The use of tetrabutylammonium fluoride to promote N- and O-(11) C-methylation reactions with iodo[(11) C]methane in dimethyl sulfoxide. | Kikuchi, T., et al. 2013. J Labelled Comp Radiopharm. 56: 672-8. PMID: 25196029
- Synthesis of alkynes from vinyl triflates using tetrabutylammonium fluoride. | Okutani, M. and Mori, Y. 2015. Chem Pharm Bull (Tokyo). 63: 393-6. PMID: 25948333
- Direct Synthesis of α-Allenols from TMS-Protected Alkynes and Aldehydes Mediated by Tetrabutylammonium Fluoride. | Huang, X. and Bugarin, A. 2016. Chemistry. 22: 12696-700. PMID: 27401054
- Electrochemical Fluorination and Radiofluorination of Methyl(phenylthio)acetate Using Tetrabutylammonium Fluoride (TBAF). | Balandeh, M., et al. 2017. J Electrochem Soc. 164: G99-G103. PMID: 28890550
- Characterization of Non-Derivatized Cellulose Samples by Size Exclusion Chromatography in Tetrabutylammonium Fluoride/Dimethylsulfoxide (TBAF/DMSO). | Rebière, J., et al. 2017. Molecules. 22: PMID: 29144402
- Mechanochemical esterification of waste mulberry wood by wet Ball-milling with tetrabutylammonium fluoride. | Chen, J., et al. 2019. Bioresour Technol. 285: 121354. PMID: 31004953
- Dependence of deposition method on the molecular structure and stability of organosilanes revealed from degrafting by tetrabutylammonium fluoride. | Miles, J., et al. 2020. Phys Chem Chem Phys. 22: 658-666. PMID: 31829362
- Fluoride-Triggered Self-Degradation of Poly(2,4-disubstitued 4-hydroxybutyric acid) Derivatives. | Kan, XW., et al. 2021. Macromol Rapid Commun. 42: e2100169. PMID: 34028933
- CO2 conversion to formamide using a fluoride catalyst and metallic silicon as a reducing agent. | Wang, R., et al. 2022. Commun Chem. 5: 150. PMID: 36698012
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetrabutylammonium Fluoride Hydrate, 25 g | sc-251153 | 25 g | $73.00 | |||
Tetrabutylammonium Fluoride Hydrate, 500 g | sc-251153A | 500 g | $243.00 | |||
Tetrabutylammonium Fluoride Hydrate, 1 kg | sc-251153B | 1 kg | $486.00 |
