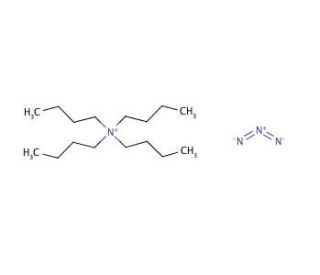
Tetrabutylammonium azide (CAS 993-22-6)
See product citations (1)
QUICK LINKS
Tetrabutylammonium azide functions as a source of azide ions in various experimental applications. Its mechanism of action involves the release of azide ions, which can then participate in a range of chemical reactions, including the formation of azides through substitution reactions. Tetrabutylammonium Azide can also act as a phase-transfer catalyst, facilitating the transfer of azide ions between immiscible phases in certain experimental procedures. Tetrabutylammonium azide can interact at the molecular level, enabling the synthesis of various organic compounds and the modification of functional groups within complex molecules. Its ability to facilitate azide-based reactions may be useful in the development of new materials and the study of organic chemistry processes.
Tetrabutylammonium azide (CAS 993-22-6) References
- Synthesis and electrochemical study of the first tetrazolate hexanuclear rhenium cluster complex. | Szczepura, LF., et al. 2007. Chem Commun (Camb). 4617-9. PMID: 17989810
- Access to optically active 3-azido- and 3-aminopiperidine derivatives by enantioselective ring expansion of prolinols. | Cochi, A., et al. 2011. Org Lett. 13: 4442-5. PMID: 21793544
- Kinetics and mechanism of the racemic addition of trimethylsilyl cyanide to aldehydes catalysed by Lewis bases. | North, M., et al. 2012. Org Biomol Chem. 10: 4289-98. PMID: 22544042
- Synthesis of hypervalent iodonium alkynyl triflates for the application of generating cyanocarbenes. | Hyatt, IF., et al. 2013. J Vis Exp.. PMID: 24056681
- Nitride-bridged triiron complex and its relevance to dinitrogen activation. | Ermert, DM., et al. 2015. Inorg Chem. 54: 9282-9. PMID: 26052673
- Ultrasound promoted mild and facile one-pot, three component synthesis of 2H-indazoles by consecutive condensation, CN and NN bond formations catalysed by copper-doped silica cuprous sulphate (CDSCS) as an efficient heterogeneous nano-catalyst. | Soltani Rad, MN. 2017. Ultrason Sonochem. 34: 865-872. PMID: 27773314
- Metal-Free Fast Azidation by Using Tetrabutylammonium Azide: Effective Synthesis of Alkyl Azides and Well-Defined Azido-End Polymethacrylates. | Wang, CG., et al. 2019. Chemistry. 25: 13025-13029. PMID: 31389637
- Toward the Scale-Up of a Bicyclic Homopiperazine via Schmidt Rearrangement and Photochemical Oxaziridine Rearrangement in Continuous-Flow. | Brown, M., et al. 2021. Org Process Res Dev. 25: 148-156. PMID: 33679122
- Asymmetric Azidation under Hydrogen Bonding Phase-Transfer Catalysis: A Combined Experimental and Computational Study. | Wang, J., et al. 2022. J Am Chem Soc. 144: 4572-4584. PMID: 35230845
- Syntheses of Legionaminic Acid, Pseudaminic Acid, Acetaminic Acid, 8-epi-Acetaminic Acid, and 8-epi-Legionaminic Acid Glycosyl Donors from N-Acetylneuraminic Acid by Side Chain Exchange. | Siyabalapitiya Arachchige, S. and Crich, D. 2022. Org Lett. 24: 2998-3002. PMID: 35420827
- Cell surface photoengineering enables modeling of glycocalyx shedding dynamics. | Purcell, SC., et al. 2022. Chem Sci. 13: 6626-6635. PMID: 35756522
- Continuous-Flow Technology for Chemical Rearrangements: A Powerful Tool to Generate Pharmaceutically Relevant Compounds. | Alfano, AI., et al. 2023. ACS Med Chem Lett. 14: 326-337. PMID: 36923914
- Facile Attachment of Halides and Pseudohalides to Dodecaborate(2-) via Pd-catalyzed Cross-Coupling. | Al-Joumhawy, MK., et al. 2023. Molecules. 28: PMID: 37050008
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tetrabutylammonium azide, 5 g | sc-229391 | 5 g | $174.00 | |||
Tetrabutylammonium azide, 25 g | sc-229391A | 25 g | $671.00 |
