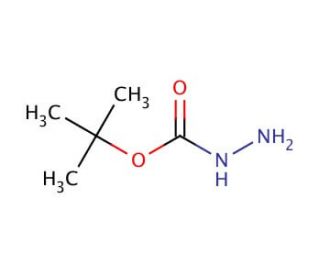
tert-Butyl carbazate (CAS 870-46-2)
QUICK LINKS
Tert-Butyl carbazate is a chemical compound that functions as a reagent in the synthesis of various organic compounds. It acts as a carbonyl-protecting group in the preparation of aldehydes and ketones. Tert-Butyl carbazate reacts with carbonyl compounds to form hydrazones, which can be further manipulated to introduce different functional groups. Tert-Butyl carbazate is particularly useful in the preparation of intermediates and agrochemicals. Its mode of action involves the formation of stable hydrazones, which can undergo subsequent transformations to yield the desired products. Tert-Butyl carbazate plays a role in the modification of carbonyl compounds, allowing for the introduction of specific chemical functionalities in a controlled manner.
tert-Butyl carbazate (CAS 870-46-2) References
- Enantiospecific synthesis of 1-azafagomine. | Ernholt, BV., et al. 2000. Chemistry. 6: 278-87. PMID: 11931107
- Stereocontrolled synthesis of triazacyclopenta[cd]pentalenes by intramolecular 1,3-dipolar cycloaddition reactions of azomethine imines. | Bélanger, G., et al. 2002. J Org Chem. 67: 7880-3. PMID: 12398520
- Palladium-catalyzed cross-coupling between vinyl halides and tert-butyl carbazate: first general synthesis of the unusual N-Boc-N-alkenylhydrazines. | Barluenga, J., et al. 2007. Org Lett. 9: 275-8. PMID: 17217283
- Synthesis and inhibitory activities of novel C-3 substituted azafagomines: a new type of selective inhibitors of α-L-fucosidases. | Moreno-Clavijo, E., et al. 2010. Bioorg Med Chem. 18: 4648-60. PMID: 20570156
- Discovery and process synthesis of novel 2,7-pyrrolo[2,1-f][1,2,4]triazines. | Thieu, T., et al. 2011. Org Lett. 13: 4204-7. PMID: 21790122
- Fluorescence turn-on detection of Sn2+ in live eukaryotic and prokaryotic cells. | Lan, H., et al. 2014. Analyst. 139: 5223-9. PMID: 25126650
- Influence of Oxidation Degree on the Physicochemical Properties of Oxidized Inulin. | Afinjuomo, F., et al. 2020. Polymers (Basel). 12: PMID: 32369991
- Continuous flow synthesis of arylhydrazines via nickel/photoredox coupling of tert-butyl carbazate with aryl halides. | Mata, A., et al. 2020. Chem Commun (Camb). 56: 14621-14624. PMID: 33151210
- Further studies on the protein conjugation of hydroxamic acid bifunctional chelating agents: group-specific conjugation at two different loci. | Safavy, A., et al. 1999. Bioconjug Chem. 10: 18-23. PMID: 9893959
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
tert-Butyl carbazate, 5 g | sc-253635 | 5 g | $23.00 | |||
tert-Butyl carbazate, 25 g | sc-253635A | 25 g | $72.00 |
