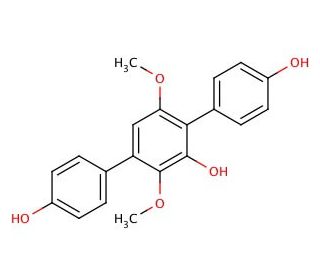
Molecular structure of Terphenyllin, CAS Number: 52452-60-5
Terphenyllin (CAS 52452-60-5)
Application:
Terphenyllin is a major polyphenyl fungal metabolites produced by Aspergillus candidus
CAS Number:
52452-60-5
Purity:
>99%
Molecular Weight:
338.4
Molecular Formula:
C20H18O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Terphenyllin is a flavanoid fungal metabolite produced de novo by Aspergillus candidus. The occurrence of this metabolite is used as a criterion in the polyphasic taxonomy of A. candidus. Terphenyllin has not been extensively studied but has been documented to exhibit activity as an anti-oxidant, a plant growth inhibitor and show weak activity against the coupled and strand transfer reaction of HIV integrase.
Terphenyllin (CAS 52452-60-5) References
- Hepato- and cardiotoxicity of xanthoascin, a new metabolite of A. candidus Link, to mice. I. Blood chemistry and histological changes in mice. | Ohtsubo, K., et al. 1976. Jpn J Exp Med. 46: 277-87. PMID: 1011375
- The protective effects of Aspergillus candidus metabolites against hydrogen peroxide-induced oxidative damage to Int 407 cells. | Yen, GC., et al. 2003. Food Chem Toxicol. 41: 1561-7. PMID: 12963009
- Isolation, structure, and HIV-1-integrase inhibitory activity of structurally diverse fungal metabolites. | Singh, SB., et al. 2003. J Ind Microbiol Biotechnol. 30: 721-31. PMID: 14714192
- Polyphasic taxonomy of Aspergillus section Candidi based on molecular, morphological and physiological data. | Varga, J., et al. 2007. Stud Mycol. 59: 75-88. PMID: 18490951
- Cytotoxic and Antibacterial Compounds from the Coral-Derived Fungus Aspergillus tritici SP2-8-1. | Wang, W., et al. 2017. Mar Drugs. 15: PMID: 29112138
- Design, semisynthesis, α-glucosidase inhibitory, cytotoxic, and antibacterial activities of p-terphenyl derivatives. | Zhang, XQ., et al. 2018. Eur J Med Chem. 146: 232-244. PMID: 29407953
- A new p-terphenyl derivative from the insect-derived fungus Aspergillus candidus Bdf-2 and the synergistic effects of terphenyllin. | Shan, T., et al. 2020. PeerJ. 8: e8221. PMID: 31915570
- Terphenyllin Suppresses Orthotopic Pancreatic Tumor Growth and Prevents Metastasis in Mice. | Zhang, J., et al. 2020. Front Pharmacol. 11: 457. PMID: 32322210
- Structure-Activity Relationships and Potent Cytotoxic Activities of Terphenyllin Derivatives from a Small Compound Library. | Haider, W., et al. 2020. Chem Biodivers. 17: e2000207. PMID: 32367656
- Metabolomic profiling of exhaled breath condensate for the diagnosis of pulmonary aspergillosis. | Wei, S., et al. 2022. Front Cell Infect Microbiol. 12: 1008924. PMID: 36159648
- Discovery of p-Terphenyl Metabolites as Potential Phosphodiesterase PDE4D Inhibitors from the Coral-Associated Fungus Aspergillus sp. ITBBc1. | Guo, Z., et al. 2022. Mar Drugs. 20: PMID: 36355001
- A Terphenyllin Derivative CHNQD-00824 from the Marine Compound Library Induced DNA Damage as a Potential Anticancer Agent. | Cao, XZ., et al. 2023. Mar Drugs. 21: PMID: 37888447
- p-Terphenyl and Diphenyl Ether Derivatives from the Marine-Derived Fungus Aspergillus candidus HM5-4. | Zeng, Y., et al. 2023. Mar Drugs. 22: PMID: 38248638
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Terphenyllin, 1 mg | sc-202358 | 1 mg | $205.00 |
