TCEP, Neutral
QUICK LINKS
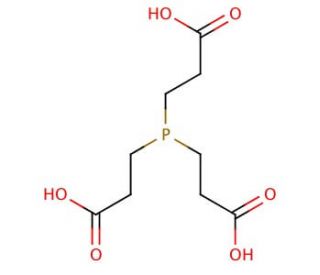
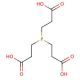
TCEP, Neutral, is a ready-to-use, stable form of the popular disulfide-reducing agent TCEP, Hydrochloride (sc-203290). TCEP, also known as tris(2-carboxyethyl)phosphine, is a neutral chemical compound widely used in various scientific and industrial applications. It is a powerful reducing agent and is particularly valued for its ability to break disulfide bonds in proteins, making it a tool in protein research and biotechnology. TCEP, Neutral′s reducing properties make it an effective tool in protein denaturation, protein structure determination, and protein purification processes. Additionally, TCEP, Neutral is highly stable and does not react with oxygen, making it suitable for use in aerobic environments. Due to its versatility and reliability, TCEP, Neutral has become a popular choice in laboratories and industries involved in protein studies, pharmaceutical development, and biotechnological advancements. Its capacity to selectively reduce disulfide bonds without interfering with other functional groups makes it an invaluable component in many biochemical and biophysical assays. TCEP′s neutral nature and compatibility with various experimental conditions make it useful in the pursuit of understanding complex biological systems and advancing scientific discoveries.
TCEP, Neutral References
- A comparison between the sulfhydryl reductants tris(2-carboxyethyl)phosphine and dithiothreitol for use in protein biochemistry. | Getz, EB., et al. 1999. Anal Biochem. 273: 73-80. PMID: 10452801
- Global phosphoproteomics identifies a major role for AKT and 14-3-3 in regulating EDC3. | Larance, M., et al. 2010. Mol Cell Proteomics. 9: 682-94. PMID: 20051463
- Identification of RhoGAP22 as an Akt-dependent regulator of cell motility in response to insulin. | Rowland, AF., et al. 2011. Mol Cell Biol. 31: 4789-800. PMID: 21969604
- Photocleavable peptide-oligonucleotide conjugates for protein kinase assays by MALDI-TOF MS. | Zhou, G., et al. 2012. Mol Biosyst. 8: 2395-404. PMID: 22772337
- Thiol-dependent recovery of catalytic activity from oxidized protein tyrosine phosphatases. | Parsons, ZD. and Gates, KS. 2013. Biochemistry. 52: 6412-23. PMID: 23957891
- Chemical synthesis of proteins using peptide hydrazides as thioester surrogates. | Zheng, JS., et al. 2013. Nat Protoc. 8: 2483-95. PMID: 24232250
- Improved production of recombinant human Fas ligand extracellular domain in Pichia pastoris: yield enhancement using disposable culture-bag and its application to site-specific chemical modifications. | Muraki, M. 2014. BMC Biotechnol. 14: 19. PMID: 24612669
- Oxidation of p53 through DNA charge transport involves a network of disulfides within the DNA-binding domain. | Schaefer, KN., et al. 2015. Biochemistry. 54: 932-41. PMID: 25584637
- Chemical synthesis of membrane proteins by the removable backbone modification method. | Tang, S., et al. 2017. Nat Protoc. 12: 2554-2569. PMID: 29189771
- Targeted Antibody Blocking by a Dual-Functional Conjugate of Antigenic Peptide and Fc-III Mimetics (DCAF). | Bai, X., et al. 2019. J Vis Exp.. PMID: 31609354
- Vimentin S-glutathionylation at Cys328 inhibits filament elongation and induces severing of mature filaments in vitro. | Kaus-Drobek, M., et al. 2020. FEBS J. 287: 5304-5322. PMID: 32255262
- Incorporating thioamides into proteins by native chemical ligation. | Fiore, KE., et al. 2021. Methods Enzymol. 656: 295-339. PMID: 34325791
- A paper-based optical sensor for the screening of viruses through the cysteine residues of their surface proteins: A proof of concept on the detection of coronavirus infection. | Gholami, MD., et al. 2022. Talanta. 248: 123630. PMID: 35660992
- Protein Profiling of a Cellular Model of NAFLD by Advanced Bioanalytical Approaches. | Altomare, AA., et al. 2022. Int J Mol Sci. 23: PMID: 36012291
- A procedure for quantitative determination of tris(2-carboxyethyl)phosphine, an odorless reducing agent more stable and effective than dithiothreitol. | Han, JC. and Han, GY. 1994. Anal Biochem. 220: 5-10. PMID: 7978256
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
TCEP, Neutral, 1 ml | sc-222338 | 1 ml | $173.00 |
