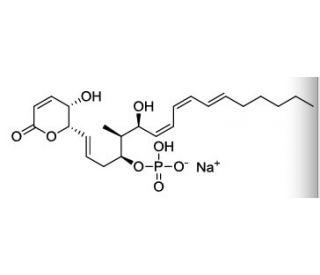
Sultriecin (CAS 131774-59-9)
QUICK LINKS
Sultriecin is a sulfur-containing antibiotic derived from the bacterium Streptomyces tendae. Its unique chemical structure features a thiotetronate motif, which is essential for its biological activity, primarily targeting bacterial protein synthesis. Sultriecin acts by binding to the 50S ribosomal subunit of bacteria, thereby inhibiting the peptidyl transferase center. This binding disrupts the elongation phase of protein synthesis, effectively preventing the formation of peptide bonds between amino acids, which is crucial for protein production. In research, sultriecin has been utilized to study bacterial resistance mechanisms and the dynamics of protein synthesis. Its ability to inhibit ribosomal function makes it a valuable tool for investigating the intricacies of ribosomal structures and functions, as well as examining how antibiotics can modulate these processes. Moreover, studies involving sultriecin have contributed to a deeper understanding of the molecular basis of antibiotic action, particularly how thiotetronate compounds interact with ribosomal components to exert their effects. This research is vital for developing new strategies to combat antibiotic resistance, providing insights that could lead to the synthesis of novel antibiotics with improved efficacy against resistant bacterial strains.
Sultriecin (CAS 131774-59-9) References
- Fostriecin: chemistry and biology. | Lewy, DS., et al. 2002. Curr Med Chem. 9: 2005-32. PMID: 12369868
- Sultriecin, a new antifungal and antitumor antibiotic from Streptomyces roseiscleroticus. Production, isolation, structure and biological activity. | Ohkuma, H., et al. 1992. J Antibiot (Tokyo). 45: 1239-49. PMID: 1399844
- Total synthesis and evaluation of cytostatin, its C10-C11 diastereomers, and additional key analogues: impact on PP2A inhibition. | Lawhorn, BG., et al. 2006. J Am Chem Soc. 128: 16720-32. PMID: 17177422
- Anti-tumor and anti-invasive effects of diverse delta-alkyllactones: dependence on molecular side-chain length, action period and intracellular uptake. | Tanaka, H., et al. 2007. Life Sci. 80: 1851-5. PMID: 17382354
- Promotive effects of hyperthermia on the cytostatic activity to Ehrlich ascites tumor cells by diverse delta-alkyllactones. | Tanaka, H., et al. 2008. Exp Oncol. 30: 143-7. PMID: 18566579
- Total synthesis, assignment of the relative and absolute stereochemistry, and structural reassignment of phostriecin (aka Sultriecin). | Burke, CP., et al. 2010. J Am Chem Soc. 132: 2157-9. PMID: 20108904
- Total synthesis and evaluation of phostriecin and key structural analogues. | Burke, CP., et al. 2010. J Org Chem. 75: 7505-13. PMID: 20669916
- Synthesis of Phostriecin and Sultriecin. | Burke, et al. 2010. Synfacts. 2010.06: 0626-0626.
- Identification of two new phosphorylated polyketides from a Brazilian Streptomyces sp. through the use of LC–SPE/NMR. | Thomasi and Sergio Scherrer, et al. 2016. Helvetica Chimica Acta. 99.4: 281-285.
- Stereoselective Synthesis of C1–C7 and C6–C22 Fragments of Phostriecin, Goniothalamines, and Their Analogues. | Purushotham Reddy, et al. 2018. European Journal of Organic Chemistry. 2018.32: 4389-4399.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sultriecin, 500 µg | sc-202350 | 500 µg | $288.00 |
