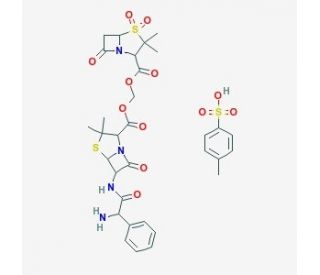
Sultamicillin tosylate (CAS 83105-70-8)
QUICK LINKS
Sultamicillin tosylate is a compound formed by the ester linkage of ampicillin and sulbactam with a tosylate group. Research on this chemical focuses on its stability and hydrolysis mechanism, exploring how it decomposes under various conditions to release its constituent molecules. Studies on sultamicillin tosylate emphasize understanding the kinetics and environmental factors affecting ester bond stability. Techniques such as chromatography and mass spectrometry are used to track the decomposition products and analyze the reaction dynamics, influenced by factors like pH levels and temperatures. The inclusion of a tosylate group in sultamicillin tosylate enhances its solubility, which is examined across various solvents. This aspect is significant for understanding how structural changes can influence physical properties like solubility. Furthermore, crystallography studies help determine its molecular configuration and interactions in the solid state. These studies are essential for developing new chemical entities and formulations that leverage the unique properties of compounds like sultamicillin tosylate.
Sultamicillin tosylate (CAS 83105-70-8) References
- Stability of sultamicillin tosylate in artificial gastric and intestinal media. | Güven, KC. and Temiz, D. 1991. Pharmazie. 46: 295. PMID: 1886922
- Radiation sterilization of anthracycline antibiotics in solid state. | Kaczmarek, A., et al. 2013. ScientificWorldJournal. 2013: 258758. PMID: 24298208
- Effect of preemptive intravenous ibuprofen on postoperative edema and trismus in third molar tooth extraction: A randomized controlled study. | Gülnahar, Y. and Kupeli, I. 2018. J Dent Anesth Pain Med. 18: 161-167. PMID: 29984320
- Treatment of Pharma Effluent using Anaerobic Packed Bed Reactor. | Vijayan, DS., et al. 2022. J Environ Public Health. 2022: 4657628. PMID: 35620734
- Ternary system of sultamicillin tosylate with hydroxypropyl-β-cyclodextrin and L-arginine: susceptibility against methicillin-resistant Staphylococcus aureus. | Bolibol, M., et al. 2022. Ther Deliv. 13: 561-575. PMID: 36861310
- Real-Life Utility of Basophil Activation Test in the Diagnosis of Immediate Hypersensitivity Drug Reactions. | Koumaki, D., et al. 2023. Dermatol Ther (Heidelb). 13: 3229-3239. PMID: 38015412
- HPLC determination of sulbactam, sultamicillin tosylate, cefaclor, ampicillin and cefoperazone in pharmaceutical preparations | El-Shanawani, A. A. 1998. Acta poloniae pharmaceutica. 55(1): 9-14.
- Radiation sensitivity and dosimetric features of sultamicillin tosylate: an electron spin resonance study | Çam, S. T., et al. 2009. Radiation Effects and Defects in Solids. 164(2): 90-100.
- Assessment of side effects of generic injectable ritodrine hydrochloride products | Gomi, Y., Matsunaga, S., Takai, Y., Fukatsu, M., Akahori, T., Ono, Y.,.. & Seki, H. 2016. Hypertension Research in Pregnancy. 4(2): 97-101.
- Antibiotic and inorganic nanoparticles co-loaded into carboxymethyl chitosan-functionalized niosome: synergistic enhanced antibacterial and anti-biofilm activities | Ashkezari, S., Abtahi, M. S., Sattari, Z., Yaraki, M. T., Hosseini, F., Salehi, R. I.,.. & Mousavi-Niri, N. 2023. Journal of Drug Delivery Science and Technology. 83: 104386.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sultamicillin tosylate, 1 g | sc-338604 | 1 g | $560.00 |
