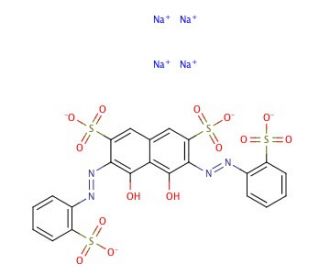
Sulfonazo III sodium salt (CAS 164581-28-6)
QUICK LINKS
Sulphonazo III sodium salt is an inorganic compound that readily dissolves in water. It possesses strong oxidizing properties and finds application in the production of certain coatings, including chrome yellow and chrome green. Additionally, this compound can be utilized for staining carbohydrates in microscopy, forming stable complexes in combination with potassium dichromate. Sulphonazo III sodium salt exhibits the ability to form stable complexes with carbon nanotubes and anions such as chloride. Its unique molecular structure allows for its concentration to be particularly high under low pH conditions and in the presence of transition metal ions. Furthermore, this compound demonstrates particle-like characteristics and exhibits an affinity for binding uronic acid molecules. Moreover, Sulphonazo III sodium salt can serve as a modifier or additive in non-aqueous electrolytes of secondary batteries.
Sulfonazo III sodium salt (CAS 164581-28-6) References
- Inhibition of tumor necrosis factor-alpha (TNF-alpha)/TNF-alpha receptor binding by structural analogues of suramin. | Mancini, F., et al. 1999. Biochem Pharmacol. 58: 851-9. PMID: 10449196
- Design and performance of a light-emitting diode detector compatible with a commercial capillary electrophoresis instrument. | Johns, C., et al. 2004. Electrophoresis. 25: 3145-52. PMID: 15472970
- Flow injection determination of formaldehyde by its catalytic effect on the oxidation of sulfonazo III by bromate with spectrophotometric detection. | Ensafi, AA. and Honarmand, E. 2005. Anal Sci. 21: 545-8. PMID: 15913145
- Azoreductase activity by purified rabbit liver aldehyde oxidase. | Stoddart, AM. and Levine, WG. 1992. Biochem Pharmacol. 43: 2227-35. PMID: 1599508
- A novel application of cyclic voltammetry for direct investigation of metabolic intermediates in microsomal azo reduction. | Zbaida, S. and Levine, WG. 1991. Chem Res Toxicol. 4: 82-8. PMID: 1912304
- The formation of an azo anion free radical metabolite during the microsomal azo reduction of sulfonazo III. | Mason, RP., et al. 1977. Biochem Biophys Res Commun. 75: 532-40. PMID: 193490
- Flow injection kinetic spectrophotometric method for the determination of trace amounts of nitrite. | Nouroozi, S. and Mirshafian, R. 2009. Talanta. 79: 1149-53. PMID: 19615524
- A rapid bioassay for chemicals that induce pro-oxidant states. | Knobeloch, LM., et al. 1990. J Appl Toxicol. 10: 1-5. PMID: 2110585
- Redox cycling of radical anion metabolites of toxic chemicals and drugs and the Marcus theory of electron transfer. | Mason, RP. 1990. Environ Health Perspect. 87: 237-43. PMID: 2176587
- Two sites of azo reduction in the monooxygenase system. | Peterson, FJ., et al. 1988. Mol Pharmacol. 34: 597-603. PMID: 2845254
- Studies on microsomal azoreduction. N,N-dimethyl-4-aminoazobenzene (DAB) and its derivatives. | Levine, WG. 1985. Biochem Pharmacol. 34: 3259-64. PMID: 3929790
- Nuclear magnetic resonance studies of the solution chemistry of metal complexes. Determination of formation constants of methylmercury complexes of selected carboxylic acids. | Libich, S. and Rabenstein, DL. 1973. Anal Chem. 45: 118-24. PMID: 4682281
- Reduction of nitro and azo compounds by NADPH-cytochrome P-450 reductase-cytochrome c heme peptide system. | Masuda, Y. and Ozaki, M. 1993. Biol Pharm Bull. 16: 112-5. PMID: 8364444
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfonazo III sodium salt, 5 g | sc-229348 | 5 g | $156.00 | |||
Sulfonazo III sodium salt, 25 g | sc-229348A | 25 g | $555.00 |
