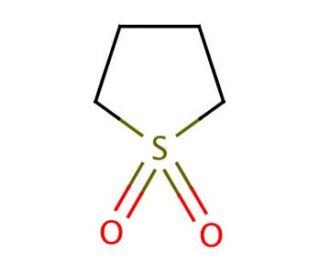
Sulfolane (CAS 126-33-0)
QUICK LINKS
Sulfolane is a high-boiling polar aprotic solvent that is and development applications. It functions as a solvent for chemical reactions and is particularly useful for extracting aromatic hydrocarbons from hydrocarbon mixtures. Its mode of action involves its ability to dissolve a wide range of organic and inorganic compounds, making it a versatile for various experimental procedures. Sulfolane′s molecular structure allows it to interact with a variety of substances, facilitating the separation and purification of compounds in laboratory applications. Its solvent properties enable it to participate in a range of chemical processes, making it a component in experimental protocols. sulfolane plays a significant role in facilitating the manipulation and isolation of various compounds in and development .
Sulfolane (CAS 126-33-0) References
- Population Pharmacokinetics of Busulfan and Its Metabolite Sulfolane in Patients with Myelofibrosis Undergoing Hematopoietic Stem Cell Transplantation. | Dadkhah, A., et al. 2022. Pharmaceutics. 14: PMID: 35745718
- Difluoro(oxalato)borate's Role in the Intercalation Behavior of Mixed Anions from Sulfolane. | Wang, Y., et al. 2023. ChemSusChem. 16: e202201218. PMID: 36039804
- Theoretical Investigation of the Structure and Physicochemical Properties of Alkaline and Alkaline Earth Metal Perchlorate Solutions in Sulfolane. | Yusupova, AR., et al. 2022. J Phys Chem B. 126: 7676-7685. PMID: 36149734
- Negative Electrospray Supercharging Mechanisms of Nucleic Acid Structures. | Ghosh, D., et al. 2022. Anal Chem. 94: 15386-15394. PMID: 36288105
- Kinetic model for the dehydration of xylose to furfural from a boronate diester precursor. | Ricciardi, L., et al. 2022. RSC Adv. 12: 31818-31829. PMID: 36380937
- Energy, exergy, economic, environment, exergo-environment based assessment of amine-based hybrid solvents for natural gas sweetening. | Ellaf, A., et al. 2023. Chemosphere. 313: 137426. PMID: 36470356
- Comprehensive H2 O Molecules Regulation via Deep Eutectic Solvents for Ultra-Stable Zinc Metal Anode. | Li, M., et al. 2023. Angew Chem Int Ed Engl. 62: e202215552. PMID: 36536537
- Does Li-ion transport occur rapidly in localized high-concentration electrolytes? | Watanabe, Y., et al. 2023. Phys Chem Chem Phys. 25: 3092-3099. PMID: 36621826
- Effects of biodegradation, biotoxicity and microbial community on biostimulation of sulfolane. | Chang, SH., et al. 2023. Chemosphere. 319: 138047. PMID: 36739988
- Cage Dynamics-Mediated High Ionic Transport in Li-O2 Batteries with a Hybrid Aprotic Electrolyte: LiTFSI, Sulfolane, and N,N-Dimethylacetamide. | Dhananjay,. and Mallik, BS. 2023. J Phys Chem B. 127: 2991-3000. PMID: 36960946
- Sulfolane-containing aqueous electrolyte solutions for producing efficient ampere-hour-level zinc metal battery pouch cells. | Wang, Y., et al. 2023. Nat Commun. 14: 1828. PMID: 37005392
- Liquid-Phase Dehydration of Glycerol to Acrolein with ZSM-5-Based Catalysts in the Presence of a Dispersing Agent. | Huang, L., et al. 2023. Molecules. 28: PMID: 37110548
- Microwave-Accelerated McKenna Synthesis of Phosphonic Acids: An Investigation. | Mustafa, D., et al. 2023. Molecules. 28: PMID: 37110732
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfolane, 5 g | sc-251090 | 5 g | $23.00 | |||
Sulfolane, 100 g | sc-251090A | 100 g | $21.00 |
