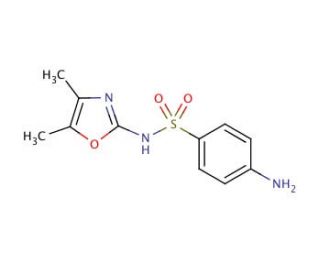
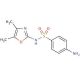
Sulfamoxole (CAS 729-99-7)
QUICK LINKS
Sulfamoxole, also known as oxasulfa or sulphamoxolum, is classified as a sulfonamide antibiotic. When used alone, it exhibits limited or no significant activity against bacteria. However, when combined with trimethoprim, it acts synergistically and demonstrates efficacy against various bacteria in laboratory applications. Notably, concentrations of 1.95-7.8 µg/ml for S. pyogenes, 0.12-1.95 µg/ml for E. coli, and 0.48-0.97 µg/ml for P. vulgaris have shown favorable results. Sulfamoxole belongs to the aminobenzenesulfonamides class of organic compounds, characterized by the presence of a benzenesulfonamide moiety with an attached amine group on the benzene ring. Structurally, sulfamoxole is composed of 4-aminobenzenesulfonic acid and 4,5-dimethyl-1,3-oxazol-2-amine, which combine to form the sulfonamide bond. It serves as both an antimicrobial agent and a drug allergen. Specifically, sulfamoxole falls into the sulfonamide, oxazole, and sulfonamide antibiotic categories.
Sulfamoxole (CAS 729-99-7) References
- Hydrogen bonding in sulfonamides. | Adsmond, DA. and Grant, DJ. 2001. J Pharm Sci. 90: 2058-77. PMID: 11745765
- Photochemical fate of sulfa drugs in the aquatic environment: sulfa drugs containing five-membered heterocyclic groups. | Boreen, AL., et al. 2004. Environ Sci Technol. 38: 3933-40. PMID: 15298203
- Characterization of sulfonamides by flow injection and liquid chromatography-electrospray ionization-mass spectrometry after online photoderivatization. | Numan, A. and Danielson, ND. 2004. J Chromatogr Sci. 42: 509-15. PMID: 15768836
- Evaluation of a method for assaying sulfonamide antimicrobial residues in cheese: hot-water extraction and liquid chromatography-tandem mass spectrometry. | Berardi, G., et al. 2006. J Agric Food Chem. 54: 4537-43. PMID: 16786995
- Selection of cytotoxicity markers for the screening of new chemical entities in a pharmaceutical context: a preliminary study using a multiplexing approach. | Gerets, HH., et al. 2009. Toxicol In Vitro. 23: 319-32. PMID: 19110050
- Comet assay with gill cells of Mytilus galloprovincialis end point tools for biomonitoring of water antibiotic contamination: Biological treatment is a reliable process for detoxification. | Mustapha, N., et al. 2016. Toxicol Ind Health. 32: 686-93. PMID: 24215064
- Synthesis, characterization, microbiological evaluation, genotoxicity and synergism tests of new nano silver complexes with sulfamoxole: X-ray diffraction of [Ag2(SMX)2]·DMSO. | Velluti, F., et al. 2014. J Inorg Biochem. 141: 58-69. PMID: 25217718
- Impact of manure-related DOM on sulfonamide transport in arable soils. | Zhou, D., et al. 2016. J Contam Hydrol. 192: 118-128. PMID: 27450276
- In vitro activity of sulfonamides and sulfones against Leishmania major promastigotes. | Peixoto, MP. and Beverley, SM. 1987. Antimicrob Agents Chemother. 31: 1575-8. PMID: 3435106
- In vitro studies with combinations of sulfamoxole and trimethoprim (co-trifamole). | Grüneberg, RN. 1982. Curr Med Res Opin. 8: 128-33. PMID: 7105824
- Reassessment of the rationale for the combinations of sulphonamides with diaminopyrimidines. | Brumfitt, W. and Hamilton-Miller, JM. 1993. J Chemother. 5: 465-9. PMID: 8195839
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfamoxole, 250 mg | sc-236952 | 250 mg | $240.00 | |||
Sulfamoxole, 1 g | sc-236952A | 1 g | $500.00 |
