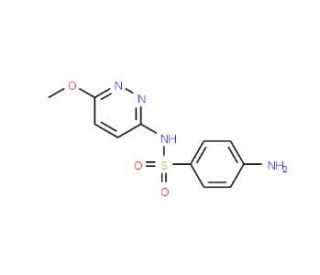
Sulfamethoxypyridazine (CAS 80-35-3)
QUICK LINKS
Sulfamethoxypyridazine is a chemical compound belonging to the sulfonamide family, primarily used in non-clinical research settings to investigate its antibacterial mechanism of action. The compound inhibits dihydropteroate synthase, an enzyme crucial for the bacterial synthesis of folic acid, which is essential for bacterial growth and replication. By interfering with this enzymatic pathway, sulfamethoxypyridazine effectively halts the production of folic acid, leading to the inhibition of bacterial proliferation. This specific inhibition is particularly fascinating for researchers because it targets a metabolic pathway unique to bacteria, offering a pathway to study selective antibacterial strategies without affecting human cells that do not synthesize their own folic acid. In scientific studies, sulfamethoxypyridazine has been extensively analyzed to understand its pharmacokinetics, resistance formation, and its impact on different bacterial species. These investigations help explain how such compounds can influence bacterial growth dynamics and contribute to the broader field of antibacterial drug development, providing insights into potential methods for overcoming bacterial resistance and enhancing the efficacy of sulfonamide drugs in controlling bacterial infections.
Sulfamethoxypyridazine (CAS 80-35-3) References
- Monoclonal antibody-based fluorescence polarization immunoassay for sulfamethoxypyridazine and sulfachloropyridazine. | Wang, Z., et al. 2007. J Agric Food Chem. 55: 6871-8. PMID: 17661485
- Photolysis of sulfamethoxypyridazine in various aqueous media: aerobic biodegradation and identification of photoproducts by LC-UV-MS/MS. | Khaleel, ND., et al. 2013. J Hazard Mater. 244-245: 654-61. PMID: 23183348
- Predicting the solubility of sulfamethoxypyridazine in individual solvents. I: Calculating partial solubility parameters. | Bustamante, P., et al. 1989. J Pharm Sci. 78: 567-73. PMID: 2778657
- Predicting the solubility of sulfamethoxypyridazine in individual solvents. II: Relationship between solute-solvent interaction terms and partial solubility parameters. | Martin, A., et al. 1989. J Pharm Sci. 78: 672-8. PMID: 2778673
- Quantum chemical investigation on photodegradation mechanisms of sulfamethoxypyridazine with dissolved inorganic matter and hydroxyl radical. | Shah, S. and Hao, C. 2017. J Environ Sci (China). 57: 85-92. PMID: 28647269
- Aqueous chlorination of sulfamethazine and sulfamethoxypyridazine: Kinetics and transformation products identification. | Nassar, R., et al. 2018. J Mass Spectrom. 53: 614-623. PMID: 29672996
- Adsorption/desorption of sulfamethoxypyridazine and enrofloxacin in agricultural soils. | Álvarez-Esmorís, C., et al. 2020. Sci Total Environ. 706: 136015. PMID: 31855641
- Environmental relevance of adsorption of doxycycline, enrofloxacin, and sulfamethoxypyridazine before and after the removal of organic matter from soils. | Alvarez-Esmorís, C., et al. 2021. J Environ Manage. 287: 112354. PMID: 33735681
- Influence of pH on the adsorption-desorption of doxycycline, enrofloxacin, and sulfamethoxypyridazine in soils with variable surface charge. | Álvarez-Esmorís, C., et al. 2022. Environ Res. 214: 114071. PMID: 35995223
- Residues of sulfadimidine/sulfanilamide and sulfamethoxypyridazine in sheep tissue. | Yndestad, M. and Underdal, B. 1977. Acta Vet Scand. 18: 15-22. PMID: 848402
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfamethoxypyridazine, 5 g | sc-251086 | 5 g | $58.00 | |||
Sulfamethoxypyridazine, 25 g | sc-251086A | 25 g | $160.00 |
