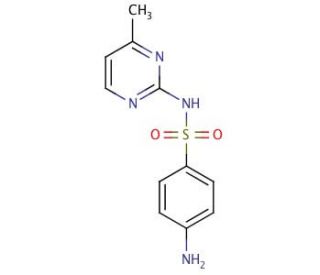
Sulfamerazine (CAS 127-79-7)
QUICK LINKS
Sulfamerazine, a chemical compound, has been a subject of extensive scientific research primarily due to its broad-spectrum antimicrobial activity against various bacterial pathogens. Its mechanism of action involves competitive inhibition of dihydropteroate synthase (DHPS), an enzyme crucial for bacterial folate synthesis. By mimicking the substrate para-aminobenzoic acid (PABA), sulfamerazine competes with PABA for the active site of DHPS, ultimately disrupting the synthesis of tetrahydrofolate, an essential precursor for DNA and RNA synthesis in bacteria. This inhibition leads to impaired nucleic acid synthesis and subsequent bacterial growth inhibition. In research applications, sulfamerazine has been utilized as a tool to study bacterial folate metabolism, enzyme kinetics, and mechanisms of antibiotic resistance. Moreover, it has been investigated for its potential synergistic effects in combination therapy regimens with other antibiotics, aiming to enhance antimicrobial efficacy and overcome bacterial resistance mechanisms. Additionally, research efforts have focused on optimizing sulfamerazine derivatives to improve its pharmacokinetic properties and broaden its spectrum of antimicrobial activity, contributing to advancements in antimicrobial research and the development of novel antibiotics.
Sulfamerazine (CAS 127-79-7) References
- Crystallization and transitions of sulfamerazine polymorphs. | Zhang, GG., et al. 2002. J Pharm Sci. 91: 1089-100. PMID: 11948548
- The influence of co-formers on the dissolution rates of co-amorphous sulfamerazine/excipient systems. | Gniado, K., et al. 2016. Int J Pharm. 504: 20-6. PMID: 26992818
- Dynamics of oxytetracycline, sulfamerazine, and ciprofloxacin and related antibiotic resistance genes during swine manure composting. | Cheng, D., et al. 2019. J Environ Manage. 230: 102-109. PMID: 30278273
- Sulfamerazine Schiff-base complex intercalated layered double hydroxide: synthesis, characterization, and antimicrobial activity. | Barnabas, MJ., et al. 2019. Heliyon. 5: e01521. PMID: 31049432
- Degradation of sulfamerazine by a novel CuxO@C composite derived from Cu-MOFs under air aeration. | Chen, Y., et al. 2021. Chemosphere. 280: 130678. PMID: 33971422
- In situ discrimination of polymorphs and phase transformation of sulfamerazine using quartz crystal microbalance. | Liu, LS., et al. 2022. Anal Chim Acta. 1221: 340137. PMID: 35934408
- Electrochemical-enhanced MoS2/Fe3O4 peroxymonosulfate (E/ MoS2/Fe3O4/PMS) for degradation of sulfamerazine. | Zhao, QM., et al. 2022. Chemosphere. 307: 136198. PMID: 36030935
- The characteristics of CDOM structural composition and the effect on indirect photodegradation of sulfamerazine. | Bai, Y., et al. 2023. Sci Total Environ. 888: 164231. PMID: 37201832
- On Interactions of Sulfamerazine with Cyclodextrins from Coupled Diffusometry and Toxicity Tests. | Sofio, SPC., et al. 2024. Biomolecules. 14: PMID: 38672478
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfamerazine, 1 g | sc-212974 | 1 g | $265.00 |
