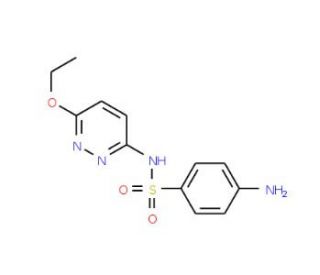
Sulfaethoxypyridazine (CAS 963-14-4)
QUICK LINKS
Sulfaethoxypyridazine is a compound that functions as a competitive inhibitor of dihydropteroate synthase, an enzyme involved in the folate synthesis pathway in bacteria. By inhibiting this enzyme, sulfaethoxypyridazine disrupts the production of tetrahydrofolate, which is essential for the synthesis of nucleic acids. This disruption ultimately leads to the inhibition of bacterial growth and proliferation. Sulfaethoxypyridazine competes with the substrate para-aminobenzoic acid (PABA) for the active site of dihydropteroate synthase, preventing the formation of dihydropteroate. As a result, the bacteria are unable to produce the necessary folate derivatives for DNA and RNA synthesis, leading to their eventual death. In this way, sulfaethoxypyridazine exerts its mechanism of action by interfering with the bacterial folate synthesis pathway, ultimately inhibiting their ability to proliferate and cause infection.
Sulfaethoxypyridazine (CAS 963-14-4) References
- Determination of sulfonamide residues in the tissues of food animals using automated precolumn derivatization and liquid chromatography with fluorescence detection. | Salisbury, CD., et al. 2004. J AOAC Int. 87: 1264-8. PMID: 15493686
- Determination of the elemental composition of trace analytes in complex matrices using exact masses of product ions and corresponding neutral losses. | Kaufmann, A. 2007. Rapid Commun Mass Spectrom. 21: 2003-13. PMID: 17546657
- Effect of water deprivation and forced exercise on blood concentrations of sulfaethoxypyridazine after its oral administration to calves. | Heath, GE. and Teske, RH. 1973. J Am Vet Med Assoc. 163: 749-52. PMID: 4777299
- Bacterial resistance to chemotherapy. I. Effect of continuous feeding of chlortetracycline, sulfaethoxypyridazine, sulfaquinoxaline, and chlortetracyclie-sulfaethoxypyridazine combinations on the development of resistance by Salmonella gallinarum in chickens. | Gale, GO., et al. 1963. Avian Dis. 7: 457-66. PMID: 5888505
- Experimental studies on an isolate of Salmonella gallinarum apparently resistant to furazolidone. | Stuart, EE., et al. 1963. Avian Dis. 7: 294-303. PMID: 5888522
- Efficacy of sulfaethoxypyridazine against fowl cholera in artificially infected chickens and turkeys, and its safety in laying chickens and broilers. | Stuart, EE., et al. 1966. Avian Dis. 10: 135-45. PMID: 5963854
- Development of cataracts in dogs and rats from prolonged feeding of sulfaethoxypyridazine. | Ribelin, WE., et al. 1967. Toxicol Appl Pharmacol. 10: 557-64. PMID: 6059670
- High-pressure liquid chromatographic determination of sulfamethazine residues in beef tissues. | Seymour, D. and Rupe, BD. 1980. J Pharm Sci. 69: 701-3. PMID: 7205587
- Gas-liquid chromatographic determination of sulfamethazine in swine and cattle tissues. | Manuel, AJ. and Steller, WA. 1981. J Assoc Off Anal Chem. 64: 794-9. PMID: 7275892
- Determination of sulfamethazine in bovine and porcine tissues by reversed-phase liquid chromatography. | Boison, JO. and Keng, LJ. 1994. J AOAC Int. 77: 558-64. PMID: 8012200
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfaethoxypyridazine, 50 mg | sc-229345 | 50 mg | $418.00 |
